| Issue |
OCL
Volume 33, 2026
Palm and palm oil / Palmier et huile de palme
|
|
|---|---|---|
| Article Number | 8 | |
| Number of page(s) | 11 | |
| DOI | https://doi.org/10.1051/ocl/2025042 | |
| Published online | 26 février 2026 | |
Research Article
Temporal patterns of Oryctes monoceros (Olivier) (Coleoptera: Scarabaeidae: Dynastinae) in oil palm replanting: A study across three cycles☆
1
Presco PLC, Obaretin Estate, Km 22, Benin-Sapele Road, Benin City, Edo State Nigeria
2
Ghana Oil Palm Development Company Ltd. (GOPDC) Kwae Estate near Kade Kwaebibirem Municipality, Eastern Region Ghana
3
Cirad, UMR ABSys, Avenue Agropolis, Montpellier, France
4
Siat Nigeria Limited (SNL), Ubima Estate, Km 6, Elele-Owerri Road, Rivers State, Nigeria
* Corresponding author: Cette adresse e-mail est protégée contre les robots spammeurs. Vous devez activer le JavaScript pour la visualiser.
Received:
17
February
2025
Accepted:
27
November
2025
Abstract
The African oil palm Elaeis guineensis Jacquin has a commercial life span between 20–30 years after which replanting is advised. Although a sound agronomic decision, planted seedlings are at risk of potential attack and damage by adult Oryctes. While it is known that adult population – in infested re‐planted palm plantation – increases with time, specific information on their population trend is limited. Hence the population of the adult Oryctes monoceros pest in 3 years of replanting (YOR) – 2018, 2020, and 2022 – in Ubima Oil Palm Plantation, Rivers State, Nigeria, was monitored, for at least twenty‐seven months, after the planting end month (PEM). Results showed progressive increase in adult population that peaked between 24 to 26 months, then began to decline. The small standard error of the mean (SEM) obtained, showed the sample mean as a precise estimate of the population mean. The one‐way analysis of variance (ANOVA) showed a highly statistical significance across the study months. Similarly, the nested ANOVA showed that variation in O. monoceros population was statistically significant across all tested variables between the three YOR. The study showed that the developmental continuity of O. monoceros is dependent on the trunk and its speed of decay. Furthermore, a second replanting phase will reach peak population faster and will surfer more damage than a first replanting phase. This study provides plantation owners and managers with information on how long the challenge of O. monoceros will likely last, as well as how best to plan replanting activities across multiple years.
Résumé
Le palmier à huile d'origine africaine Elaeis guineensis Jacquin a une durée de vie économique de 20 à 30 ans, durée à partir de laquelle il est recommandé de le replanter. Bien qu'il s'agisse d'une décision agronomique, la replantation des jeunes plants est exposée aux attaques d'Oryctes et aux dégâts associés provoqués par les imagos. Il est établi que les populations d'Oryctes augmentent avec le temps dans le cas de replantation de palmiers dans une zone déjà infestée mais il existe peu d'informations sur la dynamique de ces populations après replantation. Les populations d'imagos d’O. monoceros ont été échantillonnées pendant au moins 27 mois dès le mois de fin de plantation (MFP) dans trois replantations de palmier à huile (années de replantation (ADR): 2018, 2020 et 2022) sur la plantation d'Ubima dans l'Etat de Rivers au Nigéria. Les résultats montrent une augmentation progressive de la population d'imagos qui atteint un pic entre 24 et 26 mois avant de diminuer. La faible erreur standard de la moyenne (ESM) montre que la moyenne de l’échantillon est une estimation précise de la moyenne de la population. L'analyse de variance unidimensionnelle (ANOVA) montre une significativité statistique élevée pendant la période d’étude. Également, l'ANOVA imbriquée montre que la variation de la population d’O. monoceros est statistiquement significative pour toutes les variables testées entre les trois ADR. L’étude montre que le développement continu d'Oryctes dépend de la vitesse de décomposition du stipe. De plus, une deuxième phase de replantation aura pour conséquence d'atteindre un pic de population plus rapidement que lors d'une première phase de replantation, avec des dégâts importants. Cette étude fournit des informations aux propriétaires et gestionnaires de plantation quant au problème posé par O. monoceros en conditions de replantation et sur la meilleure stratégie de replantation à adopter sur plusieurs années.
Key words: Oil palm / replanting cycle / Oryctes monoceros / temporal patterns
Mots clés : Palmier à huile / cycle de replantation / Oryctes monoceros / patrons temporels
Contribution to the Topical Issue “Palm and palm oil / Palmier et huile de palme”.
© S.O. Riley et al., Published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Highlights
Oryctes monoceros shows distinct temporal pattern in replanting conditions.
Development and buildup of O. monoceros depends on trunk decay.
A second replanting reaches peak populations fasters than a first replanting.
Oryctes monoceros reaches peak population 24–26 months after planting ends.
1. Introduction
The African oil palm Elaeis guineensis Jacquin is typically a large perennial tree crop that may reach a height of 20 meters (Ripin and Latip, 2021). It has a productive, commercial life span of between 20–30 years (Isamail and Mamat, 2002; Syahza et al., 2018; Ashton‐Butt et al., 2019; Abubakar et al., 2023), after which harvesting becomes increasingly difficult, yield slowly declines (Corley and Tinker, 2016; Ashton‐Butt et al., 2019), thereby making it economically untenable (Hormaza et al., 2012; Abubakar et al., 2023). A sound agronomic decision involves the felling of the palm trees, and the replanting with high‐quality seedlings (Syahza et al., 2018; Ishak et al., 2020; Sembiring et al., 2021; Abubakar et al., 2023).
Replanting activities, though encouraged in old plantations, comes with risk (Syahza et al., 2018, Pradipta et al., 2020) of the potential attack and damage of planted seedlings by adult Oryctes (Giblin‐Davis, 2001; Hisham et al., 2013). Two species – Oryctes monoceros (Olivier) (Coleoptera: Scarabaeidae: Dynastinae), which is distributed throughout Africa, and Oryctes rhinoceros (Linnaeus) (Coleoptera: Scarabaeidae: Dynastinae) which is predominant in Southern and Southeast Asia (Giblin‐Davis, 2001; Philippe and Dery, 2004; EPPO, 2021; Aidoo et al., 2022a, 2022b) and the western Pacific Island (Giblin‐Davis, 2001) – are the most economically important and constitute the greatest threat to the palm industry (Manjeri et al., 2014; Aidoo et al., 2022a). For example, O. monoceros have been implicated as causing up to 40% damage to coconut and other palms in tropical Africa (Allou et al., 2006). In Nigeria, it has been documented as a major pest of palms (Agwu, 1983, Aisagbonhi, 1989, Aisagbonhi, 1994). Serious damage to oil palm plantation in Malaysia due to O. rhinoceros have been well documented. The potential of the pest to cause more than 15% reduction in canopy size (Samsudin et al., 1993), and the resultant effect on reduced photosynthetic activity, delayed plant maturity, reduced bunch size, have been estimated to cause approximately 25% crop loss (Liau and Ahmad, 1991). In severe cases, crop loss could reach between 40% to 92% during the first year of harvesting (Chung et al., 1999; Manjeri et al., 2014).
Oryctes monoceros and O. rhinoceros are holometabolous insects – undergoes complete metamorphosis (Aidoo et al., 2022a) – with morphological and biological similarities (Allou et al., 2006). The adults deposit whitish‐brown eggs (Giblin‐Davis, 2001) into rotten logs, compost and/or decaying vegetation and subsequently hatch into the larval stage after about 12–14‐days (Mariau et al., 1981; Giblin‐Davis, 2001). The larvae develop in decaying matter and navigate through three instar stages for a period of about 3 to 4 months (Mariau et al., 1981; Giblin‐Davis, 2001) depending on prevailing conditions. During development, the larvae basically act as decomposers and could be considered as beneficial (Giblin‐Davis, 2001). The third instar larvae go through an 8–13‐days prepupal stage before pupating. The pupae last for between 17–30 days, after which it emerges as adults (Mariau et al., 1981; Giblin‐Davis, 2001). The entire life cycle can last from 4 to 9 months, allowing more than one generation per year (Giblin‐Davis, 2001). While it is the larval stage that cause damage in most holometabolous insect pest of economic plants, it is the adult – specifically the young adult – that cause economic damage in O. monoceros (Giblin‐Davis, 2001; Pradipta et al., 2020). Their feeding activity – in addition to creating entry for lethal diseases (Gries and Gries, 1994) and other secondary pest (Gries and Gries, 1994; Syahza et al., 2018) such as Rhynchophorus phoenicis (Manjeri et al., 2014) – can retard the development or even cause the death of the young palms (Turner, 1981; Mariau, 2000; Aidoo et al., 2022a).
Few studies have been conducted on the population pattern of rhinoceros beetles. For example, Norman and Mohd Basri (2004) reported that the population of O. rhinoceros fluctuated with time in a replanted area, while Ripin and Latip (2021), noted that O. rhinoceros population tends to increase as the months go by after palm‐felling. Mariau et al. (1981) revealed that the attack, threatening the life of young palms by O. monoceros becomes less severe after the second year. However, specific study on the temporal pattern of adult Oryctes addressing the onset of occurrence, length of time it takes for adult O. monoceros to naturally reach peak population, and by implication peak activity, and from where the population begins to decline, in a replanted oil palm plantation have not been addressed. Although Hisham et al. (2013), showed the period when critical damage occurs in new planting, the study was on O. rhinoceros. Cik Mohd Rizuan (2014) equally studied the population dynamics of O. rhinoceros but in decomposing oil palm trunks in area practicing zero burning and partial burning.
This study monitored the population of the adult O. monoceros in three Years of Replanting (YOR) exercises – 2018, 2020, and 2022, – in Ubima Estate of Siat Nigeria Limited (SNL), Rivers State Nigeria. The aim was to determine how long after the planting end month (PEM) O. monoceros adult population: i) begins to express its presence and carry out its activities in the field, and ii) reaches “natural” peak population and begins to decline. With these plantation owners and managers are provided with valuable information to aid successful replanting exercise.
2. Materials and methods
2.1. Study site
Ubima Estate (Fig. 1) is a 9860.4 hectare (ha), third‐generation oil palm plantation that was established in 1955 (SIAT NIGERIA LTD, 2025). It is located at Ubima town in Ikwere Local Government Area of Rivers State Nigeria with geolocation between longitude 5°10'20.79“N, and latitude 6°55'14.59“E. Ubima oil palm plantation is situated in the heart of the tropical rainforest belt of the Niger Delta. The soils are generally gravel free with dominantly sandy to sandy‐loam texture, and a pH range of 2.40–4.50.
Rainfall typical follows the bimodal pattern obtainable in the equatorial climate zones. This is characterised by a major rainy season from March that peaks between June/July and minor rainy season from September to October/November, interspaced with two weeks break in August. The two seasons alternates with dry spells that lasts for about three to four months. Rainfall could reach 3227 mm per annum. Temperature is almost uniform with little variation throughout the year. Relative humidity is usually high with a mean range of 75%.
 |
Fig. 1 Map of Ubima Estate, showing the three years of replanting. |
2.2. Plot division of study site
The entire plantation was divided into standard plot size of 24.8 ha. The palms were planted in an equilateral triangular pattern at 9×9 metres spacing and 143 palms stands per hectare.
2.3. Information on replanting cycle
Before replanting commenced in 2018, palms within the plantation ranged from 25 to 30 yr of age. Hence, the third‐generation replanting cycle (RC) in Ubima estate commenced in June and ended in August 2018, with total of 899 hectares (ha) replanted. The 2018 replanting was followed by another replanting exercise in 2020, where a total of 1,079 ha was replanted. The replanting exercise commenced in April and ended in July 2020. The 2022 replanting covered 1,850.15 ha. This exercise equally commenced in April and ended in July 2022.
3. Data collection
Data on the population of the adult O. monoceros was collected through the already established planation Early Warning System (EWS). This process involved: the threshold specification for decision making, the manual collection of adults and (chemical) control activity.
3.1. Threshold specification for decision making
The breakage of at most four of the youngest eight fronds within the first rank of the oil palm phyllotaxis, in not more than 10% of palms within a plot was used as cue to initiate both manual collection of adult and chemical control. Chemical control was implemented when the threshold value obtained from the manual collection of the adults reached or exceeded a lower limit of 5 and an upper limit of 10 adults per ha.
3.2. Manual collection of adult
Data on the temporal pattern for O. monoceros adult in the three YOR were obtained from a form of adult control census. This was executed using a device‐assisted manual hand‐picking method. A 30 cm long metal rod with the tip, angled at 45° degrees (Philippe and Dery, 2004; Aidoo et al., 2022b) was used to remove the adults from within the spear where they typically cause damage. All palms within all infested plot in each YOR were observed. Both live and dead adult beetles within the spear and the surroundings of infested palms were removed and recorded. Manual collection of the adult was done either before, or after chemical application.
The data collection process was standardised in “Rounds”; a Round being interpreted as the time it takes to monitor all plots within YOR, after which monitoring returns to the starting plot. The adult control census was on a bi‐weekly round schedule, with each plot subjected to two rounds of manual collection of the adult Oryctes to reduce collection bias and improve detection efficiency.
3.3. Control activities
Chemical control was implemented in cases where damage was severe, and threshold level was exceeded. Cypermethrin (C22H19C12NO3) 10% EC, a synthetic pyrethroid insecticide was applied at 0.03% concentration; on a fortnightly schedule (Ismail et al., 2023). Although on a bi‐weekly schedule, subsequent chemical application in an infested plot was informed from the result gotten from a previous adult control census. Application of the chemical on the centre spear was by a 16 Liter knapsack sprayer at an average of 60ml per 1.15 ha.
4. Data entry/analysis
All data were entered into Excel spreadsheet and analysed using “R” version 4.4.0 (R Core Team, 2024). The average adult index (mean value) per ha, per month across all plots, per YOR was calculated by summing the total number of adult beetles collected from all infested plots within a YOR and dividing by the total number of hectares monitored; this is expressed below:
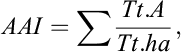
where “AAI”, is the average adult index per ha, “Tt.A”, is the total adult per plot “Tt.ha”, is the hectarage per plot and
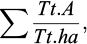
‐ is the sum of all adults per plot and the sum of each plot size (ha) as an average.
The standard error of the mean (SEM) was calculated to estimate the variability around the mean using the formular:
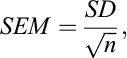
where “SD” is the standard deviation and “n” is the number of observations.
These values were used to plot a basic line graph of the pest's population trend over time, with the aid of the R package “ggplot”. Each point represents the monthly average adult population of all plots within a particular YOR. Threshold lines were included to compare the adjusted threshold adopted in this study and the recommended threshold value by Mariau et al. (1981). The PEM of each YOR was taken as the “time zero” (the reference time) from where the activity pattern of the adult population was followed.
A one‐way ANOVA was done to evaluate the effect of time – in this case months of each year – on the population trend of the adults. The month of the year was the independent variable, while the mean population per ha was the dependent variable. With this, it could be established whether the mean population per ha differed significantly across the months within each YOR; the level of significance was assessed at the 0.05 level.
A nested ANOVA to examine the variability of adult O. monoceros between the different years, and across the months was done. The various years were treated as fixed effect, with the months considered as nested within, to account for repeated temporal sampling. Both ANOVA models were implemented in R version 4.4.0 (R Core Team, 2024) using the “aov” function.
The potential effect of sample size on both F‐ and p‐ values for the ANOVA models was calculated in R version 4.4.0 (R Core Team, 2024) using the “effectsize” package (Ben‐Shachar et al., 2020). The “eta squared” (η2) analysis was estimated at 95% confidence interval.
5. Result
The temporal pattern of adult Oryctes monoceros – with the threshold lines – for the three YORs are displayed in Figures 2-4. Each data point on the line graph represents the monthly average value with the SEM as error bars, to show the precision and reliability of the sample mean compared to the population mean. The ANOVA results confirms if the observed variation in population level of the pest within and between the YORs are due to real limiting factors or just random occurrences.
 |
Fig. 2 Activity pattern of Oryctes monoceros in the 2018 YOR. |
 |
Fig. 3 Activity pattern of Oryctes monoceros in the 2020 YOR. |
 |
Fig. 4 Activity pattern of Oryctes monoceros in the 2022 YOR. |
5.1. 2018 year of replanting
The population pattern of O. monoceros adult in the 2018 replanting and how the total population per ha compare with the suggested threshold values of 15 adults/ha by Mariau et al. (1981) and the adapted threshold values of 5–10 adults per ha is displayed in Figure 2. Data collection began in November 2018, three months after the PEM, with no population of O. monoceros recorded. This occurrence continued until May 2019 – nine months after the PEM – where a total adult population of 0.02 ± 0.01 per ha was recorded. The population increased steadily, rising to 0.96 ± 0.04 adult per ha by December 2019, and 1.23 ± 0.04 in January 2020, sixteen and seventeen months after respectively. By September 2020 – twenty‐five months after the PEM – a peak population of 20.1 ± 0.59 adults per ha was reached after which the population began to decline. By September 2021, the population had dropped to 7.48 ± 0.6 adults per ha.
5.2. 2020 year of replanting
Figure 3 shows the population trend of O. monoceros in the 2020 replanting. Data collection was initiated in September 2020, two months after the planting end month, with a total population of 0.27 ± 0.08 adult per ha. An upward trend in the population was equally observed. In December 2020 a population of 0.37 ± 0.02 adults per ha was recorded, the highest for the year. The year 2021 began with a population of 0.5 ± 0.03 adults and progressively increased until June 2021 where a population of 1.19 ± 0.07 was recorded. By November, December 2021 the adult population had risen to 11.9 ± 0.4 and 10.6 ± 0.48 adults per ha respectively, with a peak population of 16.0 ± 0.83 adults per ha in January 2022 – eighteen months from the PEM. From this point onwards, the population began to decline until a population of 3.4 ± 0.45 adults per ha, was recorded in March 2023.
5.3. 2022 year of replanting
The population of adult O. monoceros in the 2022 replanting is displayed in Figure 4. Data collection commenced in August 2022, a month after the PEM with no record of the adult until October 2022 where a total population of 0.1 ± 0.12 adults per ha was recorded; this continued for the remainder of the year. While there was slight spike in population in January 2023 with a record of 1.1 ± 0.43 adult per ha, the adult population for the next five months (February to August 2023) were sub‐one values until September 2023, where a population of 1.38 ± 0.11 adults per ha was recorded. By July 2024 – twenty‐four months after the PEM, a peak population of 16.6 ± 0.55 adults per ha was reached, and from where a decline in population resulted in 1.33 ± 0.09 adults per ha as of December 2024.
5.4. Analysis of variance (ANOVA) within replanting cycles
Tables 1-3 displays result from a one‐way ANOVA conducted to evaluate the effect of time on population abundance within the three YOR. Result showed that the monthly variation in the pest population was highly statistically significant – far beyond the usual significance threshold of 0.05, 0.01, 0.001. This is expressed by large F‐ values of 652.94, 199.79 and 142.89 for the 2018, 2020 and 2022 YOR respectively and very small p‐ value of <2e‐16.
One‐way analysis of variance showing the effect of months on adult O. monoceros population density for 2018 YOR.
One‐way analysis of variance showing the effect of months on adult O. monoceros population density for 2020 YOR.
One‐way analysis of variance showing the effect of months on adult O. monoceros population density for 2022 YOR.
5.5. Analysis of variance (ANOVA) between replanting cycles
The nested ANOVA (Tab. 4) showed a highly significant effects of the two tested variables: the year of replanting (F‐value = 474.02, p‐value = <2e‐16), and the months within the various years (F‐value = 66.12, p‐value = <2e‐16) on the population levels, patterns and variability of the adult O. monoceros. This shows a clear temporal variation in the population trend of the adult across the months within each year.
Nested analysis of variance showing the effect of year, months and their interaction on adult O. monoceros population density in the three YORs.
5.6. Effect size for analysis of variance
The effect size analysis showed that month per year explained about 93% of the variance in population size (η 2 = 0.93, 95% CI [0.92, 1.00]), indicating that temporal factors exerted a dominant influence on the population trend of the pest.
6. Discussion
The study was carried out at Ubima oil palm plantation and focussed on the population trend of adult Oryctes monoceros. The aim was to identify at what point in time the increasing adult population, reaches “natural peak” and by extension exhibit a “natural decline” in numbers in post replanting conditions across multiple cycle. To achieve this, the average number of both dead and live adult O. monoceros per ha, per month and per YOR was used. This is on the rationale that whether dead or alive – with or without the influence of external factors such as weather condition or even control activities – this represents the actual number of O. monoceros adult population within the replanted plots at a particular point in time. Hence, while both manual and chemical controls were implemented, to manage the pest incidence, the aim of this study was not to primarily address efficacy of the control measures implemented.
There was no record of adult O. monoceros in plots of the 2018 replanting for the first nine months (Fig. 2). This occurrence – at face value – differ from that observed by Cik Mohd Rizuan et al. (2014) of O. rhinoceros in a replanting exercise at FELDA Global Ventures Plantation, Malaysia. However, a more in‐depth view shows an interesting similarity. According to them, O. rhinoceros was absent in the replanted plot for the first five months of observation, after which the immature stages were observed. From this account, we can infer that a felled oil palm trunk will take between 4–6 months to decay sufficiently. With the first occurrence of the adults about nine months from PEM in the 2018 plots, it would therefore justify the rationale that the decaying trunk became suitable to support the larvae from the fourth month, and by the ninth month, the first cohort of adult O. monoceros began to emerge. Thus, we can conclude that the occurrence, development and generational continuity of O. monoceros is highly dependent on the trunk and its speed of decay.
Heaps of empty fruit bunches (EFBs) unattended to, could encourage the proliferation of the pest (Hisham et al., 2013). If felled trunks or EFBs are properly managed – by say chipping of the trunk (Cik Mohd Rizuan et al., 2014; Dikrey et al., 2023) – or are prevented from natural decay, there is the possibility that their development will be significantly influenced. This conclusion agrees with Mariau and Calvez (1973), Mariau et al. (1981) Giblin‐Davis (2001), Aidoo et al. (2022b); they emphasised that the most important management option for O. rhinoceros is to ensure the destruction and elimination of the breeding sites.
Oryctes monoceros adults were already present in the 2020 RC plots barely two months after the PEM (Fig. 3). Furthermore, a population of one adult per hectare was seen six months earlier than in the 2018 RC. Though a curious occurrence, it is nevertheless expected. The adult population of O. monoceros observed in the 2020 plots must have been introduced from the neighbouring 2018 plots (Fig. 1) which had an already existing adult population. Hisham et al. (2013) noted that a second replanting phase will reach peak population faster and will suffer more damage than a first replanting phase due to an already established and multiplying beetle population from the first replanting phase. O. monoceros, though suggested to be strong fliers (Nirula, 1955, Hinckley, 1973, Monty, 1974), will remain within feeding and breeding sites. To validate this, Lever (1979), recorded a maximum flight distance of 700 m. Norman and Mohd Basri (2004) equally noted that the availability of food and abundant breeding sites in a replanted area plays a role in limiting their flight distance.
There was practically no record of O. monoceros activities in the 2022 RC plots within the first four months after the PEM (Fig. 4). Negligible adult population were recorded for the next three months, except in January 2023, where the adult population was unexpectedly high. This must have been due to the temporary termination of both monitoring and control activities within the plantation while observing the December 2022 festive break. Furthermore, two plots though planted in 2022, were located close to the 2020 plots (Fig. 1). These may have contributed to the negligible number of Oryctes adults observed in the first few months.
Peak population of O. monoceros adults was recorded at the 25th, 18th and 24th months after the PEM in the 2018, 2020 and 2022 replanting cycles respectively, followed by a steady decline. Although the period of peak population was distinct between the three replantings, it can be argued that the peak population of O. monoceros adult is best demonstrated by the expression of the pest in the 2018 and the 2022 replanting cycles. From result, we can conclude that there was no established O. monoceros adult population in the plots. Hence, every record of O. monoceros population must have been due to the replanting activity (Norman and Basri, 1997; Manjeri et al., 2014), the presence poorly managed EFBs and possible introduction from the surrounding local farms. Since the observation of the adult population in the 2020 planting was technically “fast-tracked” by eight months, it therefore means that the peak population observed, would have fallen on the 26th month mark.
The month of peak population of the adult obtained in this study, concourse with that of Hisham et al. (2013) but differs from that of Cik Mohd Rizuan et al. (2014). Hisham et al. (2013) noted two period of critical damage from Oryctes incidence in most replanted areas in FELDA Oil Pvt. Ltd., India: one within 14 to 18 months after felling and 24 to 28 months after felling, with damage in the latter significantly higher than the former. First, it is important to establish that while the result of Hisham et al. (2013) focused on critical damage, it is not implausible to conclude that the period of critical damage of Oryctes will coincide with the period of its peak population. Hence the result obtained in this study and that of Hisham et al. (2013) can be equated. There was no damage observed within the first 7–8 months of their study. Furthermore, the peak critical damage was observed between the 24 to 28 months period, like obtained in this study. As for Cik Mohd Rizuan et al. (2014) the highest population of O. rhinoceros was detected at the 10th, 13th and 16th month after felling and chipping. While the biological and morphological similarity between the two species have been emphasised, difference in geography between the two study sites as well as the uniqueness of prevailing circumstances may have caused the difference in the activity pattern of the pest's adult population between the two study sites. It should also be noted that chipping was a variable in Cik Mohd Rizuan et al. (2014) experience.
Setting threshold value is a key component of Integrated Pest Management (IPM), hence, it must be an objective reflection of the situation on ground. Manual picking of adult Oryctes served as both the major form of control and a means to quantify infestation level in plots. Hence records from this exercise and the severity of damage experienced on ground, necessitated a discretional lowering of the recommended threshold value to justify the implementation of chemical control, as well as to limit the damage and effectively manage the pest. At peak population of the adult, the economic threshold value of 15 adults Oryctes per ha, suggested by Mariau et al. (1981) was exceeded, hence chemical intervention was justified. However, this control measure would not have been applicable until the month where peak adult population was recorded. Furthermore, chemical control measures would have lasted for a few more months before and/or after the peak population month and then stopped. Based on the severity of damage on ground, it was needful to adjust the threshold value to limit the damage.
The one‐way and the nested ANOVA results provide statistical basis to validate the variability of the means based on the impacting group within and between each YOR. The one‐way ANOVA result (Tabs. 1-3) showed that the mean population value of the pest varied significantly across the months within the three replanting cycles. In other words, the result shows that the months had a highly significant effect on the population values, hence the pattern observed were therefore not just random occurrences. This is confirmed by a very large F‐value range and a very small p‐value. Similarly, the nested ANOVA result (Tab. 4) provides statistical basis to quantify the variability of the mean across the different impacting groups between the three YOR. The result showed that the mean population value of the pest varied significantly between the years of re‐planting, the months, as well as a significant interaction between the years and months.
The very large F‐ and p‐ values obtained from the ANOVA could at best indicate reaction due to large sample size or at worse exaggerated values. However, the result from the “effect size” analysis clearly indicated that monthly and yearly variation significantly influenced the population pattern during the study period and thus confirms that the observed temporal pattern is biologically meaningful and not due to a random occurrence.
A variety of chemical treatments exist for managing O. monoceros (Manjeri et al., 2014). However, Cypermethrin, a synthetic pyrethroid was used in the control of the pest when the threshold value was exceeded; it proved effective. Chung et al. (1991) noted that Cypermethrin amongst others was effective in the control of O. rhinoceros at both the nursery and in the field. Norman and Mohd Basri (1995), and Ho (1996), revealed that Cypermethrin was effective in reducing the number of holes on the spear and fronds. Although chemical intervention is justified as last resort by IPM, less environment disruptive option is advised if available. Considering this, reference is made to a success cases on the effectiveness of establishing cover cropping after felling and before replanting activities commences. Mariau and Calvez (1973), Liew and Sulaiman (1993), Ripin and Latip (2021) and Aidoo et al. (2022b) all mentioned that the planting of leguminous cover crop could act as barriers concealing rotting logs from O. monoceros, as potential breeding sites. Liew and Sulaiman (1993) noted that cover crops established to a height of 60–90 cm can hinder the breeding activity of the Oryctes beetles in a replanted area. Hence, it may not have been a coincidence that at Obaretin Estate of Presco Plc, Edo state, 524 ha and 780 ha replanted in 2018 and 2020 respectively, had minimum activities of O. monoceros adult. The geographical location between the two estates – and the various unique environmental factors – may have influenced the pests' activity. However, another difference between the two estate was the level of cover crop establishment. The cover crop plant Pueraria phaseoloides was well established in the plots planted in Obaretin estate, but not in Ubima estate. There is no hard data to prove, however, shredding/chipping of trunk, though an expensive activity was suspected to have been effective in reducing the population of O. monoceros in a replanting exercise in Ghana Oil Palm Development Company Ltd. (GOPDC) – a location with a history of O. monoceros infestation – in 2015. Biological control using Metarhizium anisopliae have been tried with relative success (Norman et al., 2005; Hisham et al., 2013; Moslim et al., 2013; Manjeri et al., 2014).
7. Conclusion and recommendation
The information from data collected in this study gives clear insight into the time when peak population density is observed in a replanted plot. The findings provides plantation owners and managers alike with information on how long the challenge of O. monoceros is likely to last if they arise from replanting activities, and the likely period (after the PEM) the pest will reach peak population and by extension the period of critical damage. A threshold range that will serve as an objective benchmark for adoption or adaptation has been suggested. A threshold population of 1 in the declining phase was observed after twenty‐seven months in the 2022 RC. It will be a useful to know exactly when the declining adult population will reach zero in subsequent related study.
The population of O. monoceros in a replanted area was low at the outset but built up once conditions in decaying felled palms trunks became favourable. Peak population was reached between 24 to 26 months after the planting end month. The proximity between the 2018 RC plots and the 2020 RC plots was a major reason behind the observation of O. monoceros adults barely two months in plots belonging to the latter YOR; the length of time the peak population was reached was also shortened. This was not the case for the 2022 YOP. This observation provides plantation owners with information on how best to plan replanting activities across multiple years. The unexpectedly high adult population in January 2023 was associated with the temporary termination of monitoring and control activities due to the December 2022 festive break. The associated damage incurred during this period is a valuable lesson on the fact that pest monitoring and management practice should continue even if pest value is below economic threshold.
Good establishment of cover crop, chipping/shredding of felled trunks and implementation of biological control were mentioned as potential alternatives for effective control of the pest. Authors such as Norman et al. (2005), Ripin and Latip (2021), have commented on the capability of cover crop to impede the activities of O. monoceros while authors such as Norman et al. (2005), Hisham et al. (2013), Moslim et al. (2013), and Manjeri et al. (2014) have commented on the efficacy of biological control. Studies on their effectiveness within the African context should be embarked on and considered for implementation going forward.
The threshold level of O. monoceros per ha that will warrant chemical intervention as suggested by Mariau et al. (1981) will need to be reviewed. This is based on the severity of damage caused by a population of ≥ 5 to ≥ 10 adults per ha, observed during the study. A threshold below the recommended number per ha should be considered, or threshold should be set on a case‐by‐case basis and determined by the severity of damage.
Acknowledgments
Special thanks to the management of Siat Nigeria Ltd for the support and for providing a conducive environment to carry out this study. We acknowledge the members of the pest monitoring team and their coordinators for the good work in ensuring and sustaining the early warning system. We thank the GIS officer for the well‐produced map.
Conflicts of interest
The authors declares that there are no conflicts of interest related to the submission and potential publication of this manuscript.
Data availability statement
Relevant data can be made available on the request of the Journal Editor‐in‐Chief.
References
- Abubakar A, Kasim S, Ishak MY, Uddin MK. 2023. Maximising oil palm yield: innovative replanting strategies for sustainable productivity. J Environ Sci 5(2): 61–75. https://doi.org/10.30564/jees.v5i2.5904. [Google Scholar]
- Agwu SI. 1983. The occurrence and breeding sites of Oryctes monoceros (Coleoptera: Dynastidae). Nigerian Institute for Oil Palm Research (NIFOR). 6: 386–399. [Google Scholar]
- Aidoo OW, Ding F, Ma T, et al. 2022a. Determining the potential distribution of Oryctes monoceros and Oryctes rhinoceros by combining machine‐learning with high‐dimensional multidisciplinary environmental variables. Sci Rep 12: 17439. https://doi.org/10.1038/s41598‐022‐21367‐1. [Google Scholar]
- Aidoo OW, Hao M, Ding F, et al. 2022b. The impact of climate change on potential invasion risk of Oryctes monoceros worldwide. Front Ecol Evol 10: 895906. [Google Scholar]
- Aisagbonhi CI, Kolade KO. 1994. A survey of insects associated with coconut palms in NIFOR, Benin with emphasis on possible vectors of Bronze leaf wilt (Awka wilt). Dis Principles 38(2): 95–99. [Google Scholar]
- Aisagbonhi CI. 1989. Insects associated with the date palm Phoenix dactylifera L. in Nigeria particular reference to pest of fruits. Unpublished PhD. Thesis, University of Benin. [Google Scholar]
- Allou K, Morin JP, Kouassi P, Nklo FH, Rochat D. 2006. Oryctes monoceros trapping with synthetic pheromone and palm material in Ivory Coast. J Chem Ecol 32: 1743–1754. [Google Scholar]
- Ashton‐Butt A, Willcock S, Purnomo D, et al. 2019. Replanting of first‐cycle oil palm results in a second wave of biodiversity loss. Ecol Evol 9: 6433–6443. [Google Scholar]
- Ben‐Shachar MS, Ludecke D, Makowski D. 2020. effectsize: Estimation of effect size incices and standardised parameters. J OpenSource Softw 5(56): 2815. [Google Scholar]
- Chung GF, Sim SC, Balasubramanian R. 1999. Effects of pest damage during immature phase on the early yields of oil palm. In: Proceeding PORIM International Palm Oil Congress. Emerging Technologies and Opportunities in next millennium, vol. 1, pp. 454–476. [Google Scholar]
- Chung GF, Sim SC, Tan MW. 1991. Chemical control of rhinoceros beetles in the nursery and immature oil palms. In: Proceedings of the PORIM International Palm Oil Development Conference – Progress, Prospect and Challenges Towards the 21st Century, vol. 1, pp. 380–395. [Google Scholar]
- Cik Mohd Rizuan ZA, Abu Hassan A, Hasber S, Noor Hisham H. 2014. Population dynamics of Oryctes rhinoceros in decomposing oil palm trunk in areas practicing zero burning and partial burning. J Oil Palm Res 26(2): 140–145. [Google Scholar]
- Corley RHV, Tinker PB. 2016. The oil palm. Chichester, UK: John Wiley and Sons, Ltd. [Google Scholar]
- Dikrey R, Gende P, Simon C, et al. 2023. Coconut Rhinoceros Beetle (CRB) control and management on oil palm in the Solomon Island. AgResearch. Poster. https://doi.org/10.57935/AGR.23916558.v1. [Google Scholar]
- European and Mediterranean Plant Protection Organisation (EPPO). 2021. EPPO global database. Paris: EPPO. [Google Scholar]
- Giblin‐Davis RM. 2001. Borers of palms. In: Howard FW, Moore D, Giblin‐Davis RM, eds. Insects on Palms. CABI Publishing, pp. 267–304. [Google Scholar]
- Gries G, Gries R. 1994. Aggregation pheromone of the African Rhinoceros Beetle, Oryctes monoceros (Olivier) (Coleoptera: Scarabaeidae). Z Naturforsch 49c: 363–366. [Google Scholar]
- Hart EA. 2016. Soil investigation for the Elele and Ubima oil plantation Estate (SIAT), Rivers State. Alex Ventures (NIG). SIAT Nigeria Limited (SNL) Archive (2016). [Google Scholar]
- Hinckley AD. 1973. Ecology of the coconut rhinoceros beetle, Oryctes rhinoceros (L.) (Coleoptera: Dynastidae). Biotropica 1973: 111–116. [Google Scholar]
- Hisham NH, Rizuan CMZA, Suhaidi H. 2013. Control measures and integrated approach for major pests of oil palm in FELDA. MPOB Palm Oil Congress (PIPOC). [Google Scholar]
- Ho CT. 1996. The integrated management of Oryctes rhinoceros (L) populations in the zero‐burning environment. In: Ariffin D, Mohd Basri W, Rajanaidu N, Mohd Tayeb D, Paranjothy K, Cheah SC, Chang KC, Ravigadevi S, eds. Proceeding of the 1996 PORIM international palm oil congress – agriculture conference. Bangi: PORIM, pp. 336–368. [Google Scholar]
- Hormaza P, Fuquen EM, Romero HM. 2012. Phenology of the oil palm interspecific hybrid Elaeis oleifera x Elaeis guineensis. Scientia Agricola 69: 275–280. https://doi.org/10.1590/S0103‐90162012 000400007. [Google Scholar]
- Ishak SM, Aman Z, Taib HM. 2020. An evaluation on outcome of oil palm replanting scheme (TSSPK) and new planting scheme (TBSPK). Int J Modern Trends Soc Sci 3(14): 129. [Google Scholar]
- Ismail A, Mamat MN. 2002. The optimal age of oil palm replanting. Oil Palm Ind Econ J 2(1): 11–18. [Google Scholar]
- Ismail IA, Zaini MR, Mazran NSS, Hasin AKM. 2023. Population's preference of rhinoceros beetle (Oryctes rhinoceros) between oil palm (Elaeis guineensis) and coconut palm (Cocus mucifera l.): Contributing to pest's controlling strategy. Asian J Agric Rural Dev 13(4): 257–268. [Google Scholar]
- Lever RJAW. 1979. Pests of the coconut palm. Rome: Food and Agricultural Organisation of the United Nations, 1969. [Google Scholar]
- Liau SS, Ahmad A. 1991. The control of Oryctes rhinoceros by clean clearing and its effect on early yield in palm‐to‐palm replants. In: Proceedings PORIM International Palm Oil Development Conference, vol. 1, pp. 396–403. [Google Scholar]
- Liew VK, Sulaiman A. 1993. The use of ground cover plants in the control of reproduction of the rhinoceros beetle (Oryctes rhinoceros) in replanting areas – current findings. Kemajuan Penyelidikan Bil 22. [Google Scholar]
- Manjeri G, Muhamad R, Tan SG. 2014. Oryctes rhinoceros Beetle, an oil palm pest in Malaysis. Annu Res Rev Biol 4(22): 3429–3439. [Google Scholar]
- Mariau D, Calvez C. 1973. Methode de lutte contre l'Oryctes en replantations de pamier a huile. Oleagineux 28: 215–218. [Google Scholar]
- Mariau D, Desmier De Chenon R, Julia Jean‐Francois, Philippe R. 1981. Oryctes monoceros (Coleoptera: Scarabeidae; Dynastinae). Oleagineux – Revue Internationale des Crops Gras. Numéro Spécial (Les Ravageurs du Palmier a Huile et du Cocotier en Afrique Occidentale). 36(4): 170–175. [Google Scholar]
- Mariau, D., 2000. Entomological problems in the replanting of oil palm and coconut plantations. Oleaginous Substances, Fats, and Lipids, 7(2), pp. 203–206. [Google Scholar]
- Monty J. 1974. Teratological effect of the virus Rhabdovirus Oryctes on Oryctes rhinoceros (L.) (Coleoptera, Dynastidae). Bull Entomol Res 64: 633–636. [Google Scholar]
- Moslim R, Kamarudin, NORMAN, Hamid, NH and Zainal Abidin, MH, 2013. Delivery techniques of Metarhizium for biocontrol of Rhinoceros Beetles in oil palm plantations. The Planter, 89(1049): 571–583. [Google Scholar]
- Nirula KK 1955. Investigation on the pest of the coconut palm II. Oryctes rhinoceros. Indian Coconut J 8: 161–180. [Google Scholar]
- Norman K, Mohd Basri W, Ramle M. 2005. Environmental factors affecting the population density of Oryctes rhinoceros in a zero‐burn oil palm replant. J Oil Palm Res 17(6): 53–63. [Google Scholar]
- Norman K, Mohd Basri W. 1995. Control method for rhinoceros beetles, Oryctes rhinoceros (L) (Coleoptera: Scarabaeidae). PORIM Occas Paper 35: 30. [Google Scholar]
- Norman K, Mohd Basri W. 1997. Status of rhinoceros beetle, Oryctes rhinoceros (Coleoptera: Scarabaeidae) as a pest of young oil palm in Malaysia. The Planter 73(850): 5–21. [Google Scholar]
- Norman K, Mohd Basri W. 2004. Immigration and activity of Oryctes rhinoceros within a small oil palm replanting area. J Oil Palm Res 16(2): 64–77. [Google Scholar]
- Philippe R, Dery SK. 2004. New way of controlling Oryctes monoceros (Coleoptera, Dynastidae), a coconut pest in Ghana. Coconut Res Dev J 20: 43–51. [Google Scholar]
- Pradipta AP, Wagiman FX, Witjaksono. 2020. The potency of collecting larvae of Oryctes rhinoceros L. (Coleoptera: Scarabaeidae) in the oil palm plantation. J Agric Sci 42(1): 153–159. [Google Scholar]
- Core Team R. 2024. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. https: //www.R‐project.org. [Google Scholar]
- Ripin MAF, Latip SNHM. 2021. Oryctes rhinoceros infestation and its interaction with oil palm damage in selected oil palm plantation. Int Trans J Eng Manag Appl Sci Technol 12(9): 1–10. [Google Scholar]
- Samsudin A, Chew PS, Mohd MM. 1993. Oryctes rhinoceros: breeding and damage on oil palm in an oil palm to oil palm replanting situation. Planter 68(813): 583–591. [Google Scholar]
- Sembiring N, Napitupulu HL, Sembiring MT, et al. 2021. The role of model simulation for replanting activities: a review. IOP Conf Ser: Mater Sci Eng 1122(1): 012055. [Google Scholar]
- SIAT NIGERIA LTD. Available from http://www.siatnigeria.com/company‐profile/brief‐history‐of‐siat‐nigeria/(last consult (2025/05/02). [Google Scholar]
- Syahza A, Bakce D, Asmit B. 2018. Increasing the awareness of palm oil plantation replanting through farmers training. Riau J Empowerment 1(1): 1–9. [Google Scholar]
- Turner PD. 1981. Oil palm diseases and disorder. Kuala Lumpur Malaysia: The Incorporated Society of Planter. p. 280. [Google Scholar]
Cite this article as:. Riley S.O. Wiafe E.K, Impens R, Beaudoin‐Ollivier L., Samuel D.O. Okoye G., Sule B.A., 2026. Temporal patterns of Oryctes monoceros (Olivier) (Coleoptera: Scarabaeidae: Dynastinae) in oil palm replanting: A study across three cycles. OCL 33: 8. https://doi.org/10.1051/ocl/2023022
All Tables
One‐way analysis of variance showing the effect of months on adult O. monoceros population density for 2018 YOR.
One‐way analysis of variance showing the effect of months on adult O. monoceros population density for 2020 YOR.
One‐way analysis of variance showing the effect of months on adult O. monoceros population density for 2022 YOR.
Nested analysis of variance showing the effect of year, months and their interaction on adult O. monoceros population density in the three YORs.
All Figures
 |
Fig. 1 Map of Ubima Estate, showing the three years of replanting. |
| In the text | |
 |
Fig. 2 Activity pattern of Oryctes monoceros in the 2018 YOR. |
| In the text | |
 |
Fig. 3 Activity pattern of Oryctes monoceros in the 2020 YOR. |
| In the text | |
 |
Fig. 4 Activity pattern of Oryctes monoceros in the 2022 YOR. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.




