| Issue |
OCL
Volume 33, 2026
Minor oils from atypical plant sources / Huiles mineures de sources végétales atypiques
|
|
|---|---|---|
| Article Number | 15 | |
| Number of page(s) | 11 | |
| DOI | https://doi.org/10.1051/ocl/2026005 | |
| Published online | 01 April 2026 | |
Review
The fatty acid and amino profiles, bioactive compounds, and alpha glucosidase inhibition activity of C. rheedei J.F. Gmelin seed oil☆
Profils des acides gras et des acides aminés, composés bioactifs et activité inhibitrice de l'α-glucosidase de l'huile de graines de C. rheedei J.F. Gmelin.
1
Laboratory of Biophysics, Institute for Advanced Study in Technology, Ton Duc Thang University, Ho Chi Minh City, Vietnam
2
Faculty of Pharmacy, Ton Duc Thang University, Ho Chi Minh City, Vietnam
3
Institute of Chemistry, Vietnam Academy of Science and Technology, 18 Hoang Quoc Viet, Nghia Do, Ha Noi, Vietnam
4
Max Rubner-Institut (MRI), Department of Safety and Quality of Cereals, Working Group for Lipid Research, Detmold 32756, Germany
5
Faculty of Biotechnology, Chemistry & Environmental Engineering, Phenikaa University, Nguyen Trac street, Ha Dong, Hanoi, Vietnam
6
Institute of Science and Technology for Energy and Environment, Vietnam Academy of Science and Technology, 18 Hoang Quoc Viet, Nghia Do, Ha Noi, Vietnam
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
22
August
2025
Accepted:
28
January
2026
Abstract
C. rheedei J.F. Gmelin is an underutilized Asian shrub with no prior characterization of its seeds or oil. This study reports, for the first time, the nutritional and functional profile of C. rheedei seeds. The seeds yielded 32.6% oil, dominated by oleic acid (71.4%) with a balanced ω-3/ω-6 ratio (∼1:1). Tocopherols (31.5 mg/100 g) and phytosterols (1388 mg/kg; β-sitosterol, stigmasterol) were abundant, supporting antioxidant and anti-inflammatory potential. Twelve free amino acids were identified, with essential amino acids comprising 22.3% of the total; proline and hydroxyproline contribute to collagen stability, while serine and alanine may provide skin-calming effects, extending relevance to cosmeceutical applications. Functionally, the oil showed strong antioxidant activity (DPPH IC50 = 13.1 μg/mL; FRAP = 2855.5 μmol Fe2+/L; TEAC EC50 = 22.8 μg/mL) and potent α-glucosidase inhibition (IC50 = 102.4 μg/mL), surpassing acarbose. Preliminary toxicology indicates no acute toxicity, though a full safety evaluation is needed. With their favorable composition and high yield, C. rheedei seeds represent a promising new economic resource for nutraceutical, functional food, and cosmetic industries in Southeast Asia.
Résumé
C. rheedei J.F. Gmelin est un arbuste asiatique sous-exploité dont les graines et l’huile n’avaient pas été caractérisées. Cette étude présente, pour la première fois, le profil nutritionnel et fonctionnel des graines de C. rheedei. Les graines ont fourni 32,6% d’huile, dominée par l’acide oléique (71,4%), avec un rapport ω-3/ω-6 équilibré (∼1:1). Les tocophérols (31.5 mg/100 g) et les phytostérols (1.388 mg/kg; β-sitostérol, stigmasterol) étaient abondants, soutenant un potentiel antioxydant et anti-inflammatoire. Douze acides aminés libres ont été identifiés, les acides aminés essentiels représentant 22.3% du total; la proline et l’Hydroxyproline contribuent à la stabilité du collagène, tandis que la sérine et l’alanine peuvent exercer des effets apaisants cutanés, conférant une pertinence cosméceutique. Fonctionnellement, l’huile a montré une forte activité antioxydante (DPPH CI50 = 13.1 μg/mL; FRAP = 2 855.5 μmol Fe2+/L; TEAC CE50 = 22.8 μg/mL) et une inhibition marquée de l’α-glucosidase (CI50 = 102.4 μg/mL), surpassant l’acarbose. Des données toxicologiques préliminaires indiquent l’absence de toxicité aiguë, bien qu’une évaluation complète reste nécessaire. Grâce à sa composition favorable et à son rendement élevé, C. rheedei constitue une nouvelle ressource économique prometteuse pour les industries nutraceutique, alimentaire fonctionnelle et cosmétique en Asie du Sud-Est.
Key words: C. rheedei / fatty acid / oleic acid / tocopherol / sterol / amino acids / antioxidant / antidiabetic activity
Mots clés : C. rheedei / acide gras / acide oléique / tocophérol / stérol / acides aminés / antioxydant / activité antidiabétique
Contribution to the Topical Issue: “Minor oils from atypical plant sources / Huiles mineures de sources végétales atypiques”.
© EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Highlights
First comprehensive study on C. rheedei seed oil composition and bioactivity.
Strong antioxidant and α-glucosidase inhibitory activity, surpassing acarbose.
Promising new source for functional foods, nutraceuticals and cosmetic products.
1 Introduction
Nutritional characterization of seeds and their oils has been a growing focus given their concurrent role as providers of fundamental nutrients and functional bioactivities. Fatty acids, tocopherols, sterols, and amino acids are key dietary ingredients that not just provide for basic nutrition but also chronic disease prevention with their antioxidant and metabolism regulatory activities (Gunstone & Harwood, 2007; Maestri et al., 2019). Natural antioxidants like tocopherol-based vitamin E shield cell structures against oxidative stress, while the phytosterols control cholesterol levels and promote metabolism equilibrium (Simopoulos, 1996; Zhang et al., 2020). Amino acids, like proline and hydroxyproline, have critical functions in collagen stability, tissue maintenance, and immune support (Kumar et al., 2016; Xu et al., 2019), so their profiling is even relevant at their presence even at moderate amounts.
C. rheedei J.F. Gmelin (Opiliaceae) is a ubiquitous Asian tropics-distributed shrub traditionally used for infectious diseases and gastrointestinal ailments (Hiepko et al., 2008; Mounnissamy et al., 2009). Even though leaves and roots have been found to show evidence of their antidiabetic and antioxidant activities (Revathi et al., 2018; Salehi et al., 2019), their seeds and oil have not been taken up for systematic studies yet. On the contrary, renowned edible oils like olive and sesame have been comprehensively characterized with respect to their fatty acid profile, tocopherols, and sterols and also attributed with glycaemic control and oxidative stress prevention, respectively (Massimo et al., 2009; Jacinta et al., 2017; Özcan et al., 2019).
Antioxidant activity is of specific concern, due to the key role that oxidative stress plays in the development of chronic diseases. Bioactive compounds from seed oils can mitigate oxidative damage, reduce inflammation, and stabilize cell membranes, thereby lowering the risk of conditions such as diabetes and cardiovascular complications (Benavente-García et al., 2000; Bendini et al., 2007). Coincidentally, α-glucosidase inhibition is a proven pathway to diminish postprandial hyperglycemia, and herbal oils that are oleic acid-rich, tocopherol-rich, and phytosterol-rich have been shown to exhibit this activity (Chen et al., 2004; Jacinta et al., 2017; Salehi et al., 2019).
Comprehensive data have not been reported yet on the seed composition of C. rheedei, not to mention its fatty acids, tocopherols, sterols, and amino acids, nor on its functional antioxidant and antidiabetic activities. To the authors' knowledge, this is the first report that gives a combined nutrition and functional analysis on C. rheedei seeds and seed oil. By integrating compositional analysis with antioxidant and α-glucosidase inhibition tests, this paper gives evidence for the novelty of exploring C. rheedei as a potential under-explored resource with potential applications in the developing world for the production of nutraceuticals and functional foods.
2 Materials and methods
2.1 Sampling
The seeds of C. rheedei J.F. Gmelin (Opiliaceae) were collected in Ba Ria-Vung Tau province, Vietnam, and identified and given a herbarium number by Dr. Nguyen Quoc Binh (Department of Biology, Vietnam National Museum of Nature, VAST). Voucher specimens VNMN-B 2024.12 were kept at the Department of Lipids and Applied Computational Chemistry, Institute of Chemistry, VAST, and stored at −4°C for further experiments. All solvents and standards (fatty acid methyl ester mix, tocopherol standards, phytosterol standards, and amino acid standard mixture) were of analytical grade and purchased from Merck (Darmstadt, Germany) or Sigma-Aldrich (St. Louis, MO, USA).
2.2 Total lipid extraction
The oil extraction from C. rheedei seeds was performed according to the ISO reference method ISO 659:2009 (Oilseeds—–determination of oil content) using a Twisselmann apparatus. Approximately 10 g of ground seeds were extracted with 75 mL of petroleum ether (Merck, Darmstadt, Germany) for 6 h. The solvent was removed with a rotary evaporator at 40°C and 25 Torr, and residual traces were evaporated under nitrogen. The oil was filtered and stored at 4°C until analysis.
2.3 Fatty acid analysis
Fatty acid methyl esters (FAMEs) were identified by comparison with a 37-component FAME Mix standard (Supelco® TraceCERT® CRM47885, Merck KGaA, Darmstadt, Germany). The fatty acid composition was determined according to the International Organization for Standardisation method ISO 5509:1988 (Animal and vegetable fats and oils—preparation of methyl esters of fatty acids). Prior to analysis, fatty acids were converted to their methyl esters. Briefly, seed oil (approximately 50 mg) was dissolved in 1 mL of n-heptane (Merck, Darmstadt, Germany). Subsequently, 50 μg of sodium methylate (Merck, Darmstadt, Germany) was added, and the mixture was stirred for 60 s at room temperature. The reaction was terminated by adding 100 μL of distilled water, and the solution was neutralized with 50 μL of 1 mol/L HCl containing methyl orange indicator (Merck, Darmstadt, Germany). The mixture was then dried with 20 mg of sodium hydrogen sulfate (Merck, Darmstadt, Germany). The upper n-heptane phase was transferred into a vial for gas chromatographic (GC) analysis.
GC analysis was performed on a Hewlett-Packard Model 5890 Series II/5989. A gas chromatograph equipped with a ZB-1 fused-silica capillary column (100 m × 0.25 mm internal diameter, 0.2 μm film thickness; Phenomenex, Torrance, CA, USA). The oven program was initial temperature of 155°C, increased to 220°C at 1.5°C/min, and held for 10 min. Injector and detector temperatures were set at 250°C. Hydrogen was used as the carrier gas (linear velocity 36 cm/s). The split ratio was 1:50. Detector gases were hydrogen (30 mL/min), air (300 mL/min), and nitrogen (30 mL/min). The injection volume was less than 1 μL (manual mode). Peak identification and quantification were performed with the instrument’s integration software, and fatty acid contents were expressed as relative percentages of total fatty acids through direct internal normalization.
2.4 Tocopherol
The determination of tocopherol was performed according to the ISO International Organization for Standardization standard method, 2016 ISO 9936:2016 (Determination of tocopherols and tocotrienols by HPLC). A 250 mg aliquot of oil was diluted in 25 mL heptane (Merck, Darmstadt, Germany). Analysis was carried out on a Merck–Hitachi low-pressure gradient HPLC system with an L-6000 pump, an F-1000 fluorescence detector (excitation 295 nm, emission 330 nm), and the integrated Chemstation software. Approximately 20 μL of the sample was injected using the Merck 655-A40 automatic sampler into a Diol phase HPLC column (inner diameter 250 mm × 4.6 mm; Merck), operated at a flow rate of 1.3 mL/min. The mobile phase used is heptane/tert-butyl methyl ether (99/1, v/v). The results are presented as mg of vitamin E/100 g of oil. (ISO 9936:2016). Quantification was calculated based on calibration curves prepared with authentic tocopherol standards (α, β, γ, and δ) expressed as mg/100 g oil. Authentic standards of α-, β-, γ-, and δ-tocopherols (Sigma–Aldrich, USA) were used.
2.5 Sterol
Sterol was determined according to the official IOOC (International Olive Oil Council, 2001) method. An oil sample (15 g) was saponified by 50 mL of 2 N ethanolic potassium hydroxide solution. The unsaponifiable fraction was dissolved in chloroform, and approximately 20 mg were loaded on a basic silica TLC plate. The sterol and triterpenediol fraction were separated by eluent mixture with hexane and diethyl ether 65:35 (v/v). The corresponding band was visualized under UV light after being sprayed with 2.7-dichlorofluorescein in a 0.2% ethanolic solution, scraped off with a spatula, and extracted with chloroform. After the extract was evaporated to dryness, sterols and triterpenediols were converted into trimethylsilyl ethers by the addition of pyridine hexamethyldisilizane-trimethylchlorosilane (9:3:1, v/v/v). The resulting mixture was kept for 15 min and then centrifuged. The analysis of sterols was performed on a fused silica capillary column coated with 2% isopropanol/98% hexane (30 m x 0.32 mm i.d., film thickness 0.25 μm; Rtx-5: Restek Corporation, Bellefonte, PA, USA; or HP-5: Agilent Technologies Inc., Little Falls, DE, USA) with a Hewlett Packard series 6890 GC (Waldbronn, Germany) equipped with a split/splitless injector, an autosampler, and a flame ionisation detector (FID). An aliquot of 1.0 mL of derivatized sample solution was introduced into the column via split injection (300°C, split ratio 1:50). The components were separated isothermally at 300°C and detected with the FID (at 310°C). The carrier gas was helium. To identify the individual peaks of sterols, the determination of relative retention times (RRT) for sterols was carried out according to the majority compound of sterols (β-sitosterol), knowing that RRT (β-sitosterol) equals 1 as described by the COI (International Olive Council, 2013). (COI/T.20/Doc. No. 10/Rev. 2001; COI/T.20/Doc. No. 30/Rev. 2013). Sterol quantification was based on comparison with authentic sterol standards, expressed as mg/kg of oil. Sterol standards (β-sitosterol, stigmasterol, campesterol, Sigma–Aldrich, USA) were used as external references.
2.6 Amino acid analysis
Free amino acids were analyzed using an Agilent 1100 High-Performance Liquid Chromatography (HPLC) system (Agilent Technologies, Santa Clara, CA, USA). Separations were performed with a cation exchange column (3 × 250 mm, 8 μm particle size; Pickering Laboratories Inc., Mountain View, CA, USA) at 40°C with a flow rate of 0.3 mL/min. The reactor was a Pinnacle PCX (Pickering Laboratories Inc.), and the temperature was 130°C. The seed was ground in a ball mill with a mass of about 0.08 g. Add 1 mL of 50% MeOH, vortex, and ultrasonicate at 50–60°C for 30 min. Transfer the vials to a thermostatic surface of 55°C (± 1°C) for 10 min. Remove to cool. Transfer the post-derivative solution to the insert and proceed with the chromatographic analysis. The mixture was diluted 40-fold using 10 μL column injections of lithium diluents (pH 2.36). The amino acid concentrations of layers were calculated from calibration curves based on amino acid standard mixtures (Pickering Laboratories Inc.) (Jaudzems et al., 2018; Salazar et al., 2012). Only free amino acids were quantified in this study. The Agilent 1100 Series HPLC system is widely applied for amino acid and tocopherol analysis (Agilent Technologies, USA).
2.7 Antioxidant assay by DPPH
The antioxidant activity of the extracts was evaluated using DPPH as reported by Saeed et al. (2012). An aliquot (0.5 mL) of the DPPH solution (about 50 mg/100 mL) was diluted in 4.5 mL of methanol, and 0.1 mL of a methanol solution of the extract was added. The mixture was shaken vigorously and allowed to stand for 45 min in the dark. The decrease in absorbance was measured at 515 nm against a blank (without extract) with a spectrophotometer. From a calibration curve obtained with different amounts of extracts, the IC50, which was required to quench 50% of the initial DPPH radicals under the experimental conditions given, was calculated.
2.8 Ferric reducing antioxidant power assay
The ferric reducing ability of the samples was evaluated according to the modified method of Worachai et al. (2023). The ferric reducing antioxidant power (FRAP) reagent contained 2,4,6-tripyridyl-s-triazine (TPTZ) solution (20 mM) in HCl (40 mM), FeCl3.6H2O (20 mM), and 0.3 M acetate buffer with pH 3.6. FRAP reagent (1.8 mL) mixed with 0.2 mL of test sample was incubated at 37°C for 10 min in a water bath. After incubation, the absorbance was measured immediately at 593 nm. The calibration curve was plotted with absorbance concentration of FeSO4 in the range of 0–1 mM, and the total antioxidant activity was expressed as μmol Fe (II)/g extract.
2.9 TEAC
The TEAC (Trolox equivalent antioxidant capacity) was evaluated according to the modified method of Yuvaraj et al. (2013). The green radical cation ABTS+ was generated by oxidation of 7 mM ABTS [2,2ʹ-azinobis(3-ethylbenzothiazoline-sulfonic-6-acid) diammonium salt] with 2.45 mM K2S2O8 and allowing the mixture to sit in the dark at room temperature for 16 h. The ABTS+ solution was diluted to an absorbance of 0.7 ± 0.02 at 734 nm. After the addition of 1900 μl of diluted ABTS+ solution to 100 μl of test substances or Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid) standards, the absorbance was taken 1–6 min after initial mixing. The percentage inhibition of absorbance of ABTS+ at 734 nm was then calculated and plotted toward concentrations of test/standard substances as a function of time or concentration. The TEAC value was finally calculated as the ratio of the slopes of the linear regression of the concentration–response curves of the test substances toward the reference substance (Trolox).
2.10 Alpha-glucosidase inhibition assay
The procedure for assessing α-glucosidase enzyme inhibition activity was conducted on a 96-well plate (Chen et al., 2004; Hakamata et al., 2016). The enzyme α-glucosidase breaks down the p-Nitrophenyl-α-D-glucopyranoside substrate, which lets off yellow p-Nitrophenol.
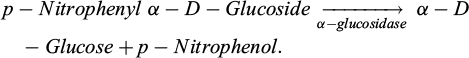
The reaction mixture’s absorbance at 410 nm half an hour after the reaction. The enzyme α-glucosidase’s activity can be measured by the amount of p-Nitrophenol it makes. We used a 96-well plate to find out how well the enzyme α-glucosidase inhibits things from happening. To make the chemical less concentrated, we combined it with DMSO and deionized water in different proportions. We mixed the test sample with DMSO and deionized water to make a range of concentrations: 256, 64, 16, and 4 μg/ml. We could also dilute it more with samples that had less activity. The reference substance was acarbose, and all assays were carried out in triplicate. The procedure had 100 mM phosphate buffer at pH 6.8, 0.2 U/ml α-glucosidase, the test sample, and 2.5 mM p-nitrophenyl α-D-glucopyranoside. The reaction buffer took the place of the test sample in the control. The experiment was kept at 37°C. Na2CO3 was used to terminate the reaction after 30 minutes. A BIOTEK reader at a wavelength of 410 nm (A) was used to find the reaction's absorbance. We used the following formula to figure out how well the test sample stopped α-glucosidase from working:
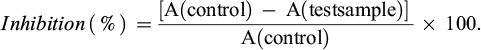
IC50 (half maximal inhibitory concentration) is the concentration of the test substance that inhibits 50% of α-glucosidase enzyme activity and was calculated using Tablecurve software.
2.11 Statistical analysis
All experiments were conducted in triplicate (n = 3). Results are expressed as mean ± standard deviation (SD). Statistical analyses were performed using SPSS Statistics 22.0 (IBM Corp., Armonk, NY, USA). One-way analysis of variance (ANOVA) followed by Tukey’s post-hoc test was applied to determine significant differences among means, with P < 0.05 considered statistically significant.
3 Results and discussion
3.1 Total lipid
Soxhlet extraction according to ISO 659:2009 revealed that C. rheedei seeds contained 32.58% oil (dw). This value is comparable to olive seeds (∼35%) (García-Inza et al., 2016), higher than soybean (18%–22%) and sunflower (20%–32%), but lower than peanut (40%–50%) and rapeseed (40%–60%) (Gunstone et al., 2007). While the seeds are traditionally consumed, food safety evaluations are limited; preliminary reports suggest no acute toxicity from extracts of C. rheedei J. Gmelin (Mounnissamy et al., 2010). The relatively high oil yield and bioactive profile highlight potential for nutraceutical and functional food applications in Southeast Asia.
Fatty acids composition
The fatty acid profile of C. rheedei seed oil was dominated by oleic acid (C18:1, 71.38%), followed by palmitic (12.11%), linoleic (5.78%), linolenic (5.73%), stearic (2.90%), and palmitoleic (2.90%). This predominance of C16 and C18 fatty acids is typical of seed oils and places C. rheedei closer to olive oil (oleic-rich) than to soybean or sunflower oils (linoleic-rich) (Massimo et al., 2009; Mostafa et al., 2021). The oil contained both omega-3 (α-linolenic) and omega-6 (linoleic) fatty acids, which together accounted for 10.74% of the total. The n-3/n-6 ratio was ∼1:1, within the recommended dietary range of 1:1–1:4 (Guillocheau et al., 2017). Saturated fatty acids comprise about 15% of the total, mainly palmitic acid.
Oleic acid is a fatty acid that is beneficial for your health since it helps with heart problems, keeps blood sugar levels stable, and fights free radicals. Studies on animals and people have indicated that a diet high in oleic acid can improve insulin work in both healthy people (Alcubierre et al., 2016) and those with diabetes (Teo et al., 2021). Oleic acid can also help oils hold onto moisture and not irritate skin and hair, which is beneficial for hair and skin care products. Fatty acid methyl esters were identified by comparison with a 37 FAME Mix standard (Supelco, USA). C. rheedei seed oil has two important omega fatty acids: omega-3 (alpha-linolenic acid) and omega-6 (linoleic acid), and they make up 10.74% of the oil. The ratio between them is 1:1. It is critical to have the appropriate balance between omega-3 and omega-6 fatty acids because too much omega-6 might cause inflammation and other health problems (Wanna et al., 2007). The ideal ratio for a healthy diet is between 1:1 and 1:4 (omega-3 to omega-6) (Etienne Guillocheau et al. 2017). C. rheedei seed oil contains the perfect ratio for human health, which is 1:1.
 |
Fig. 1 Chromatogam of fatty acids |
The fatty acids composition (%) of C. rheedei seed oils
3.2 Tocopherol
The findings in Table 2 indicate that the total tocopherol concentration in C. rheedei seed oil was 31.54 mg/100 g, comprising α-tocopherol at 21.13 mg/100 g (66.6%), γ-tocopherol at 9.26 mg/100 g, and plastochromanol-8 (P8), a tocopherol-like antioxidant compound (Kruk, 2014), at 1.15 mg/100 g. The main type of tocopherol is α-tocopherol. It is known for its important function in stopping lipid oxidation, lowering oxidative stress, and helping to control blood sugar levels. Compared to Gemlik olive oil, C. rheedei oil has more γ-tocopherol: 8.12 mg/100 g (Simopoulos et al., 2016). Wheat germ oil, on the other hand, has a lot of α-tocopherol, which is the most prevalent isomer in common vegetable oils, including barley, cottonseed, olive, palm, and sunflower, as well as in some specialty oils like grapeseed, papaya, orange, and lemon. Specifically, cashew oil is the only oil documented to have beta-tocopherol as the primary type (Özcan et al., 2019). Tocopherol works best when it is mixed with other substances, like polyphenols (oleuropein, hydroxytyrosol), flavonoids, and polyunsaturated fatty acids (PUFA). These interactions assist in boosting the effects of antioxidants and anti-inflammatories and make insulin more sensitive. For instance, tocopherol and omega-3 fatty acids together can make your metabolism work better, lower oxidative stress, and save you from getting diabetes. The oil from C. rheedei seeds is very nutritious and has many tocopherols, especially α- and γ-tocopherols. It could be used to help prevent and treat chronic diseases like diabetes.
 |
Fig. 2 Chromatography of tocopherol |
The tocopherol content (mg/100 g) of C. rheedei seed oils.
3.3 Sterol
This study focused on major lipid-related bioactives (tocopherols, sterols, and amino acids) using standardized ISO/IOOC methods. LC–MS-based profiling of minor phytochemicals was beyond the current scope and is suggested for future research.
The seeds of C. rheedei contain a total phytosterol concentration of 1388.07 mg/kg. The three most abundant phytosterols are sitosterol (790.46 mg/kg), stigmasterol (249.08 mg/kg), and sitostanol (104.28 mg/kg). The overall sterol content in these seeds is comparable to that found in common cooking oils (Gomez-Caravaca et al., 2010). These seeds also contain several types of sterols, with quantities ranging from 2.09 to 97.51 mg/kg. A significant amount of β-sitosterol and stigmasterol is present, which are important because they help lower cholesterol and promote heart health. Phytosterols and other sterols are the most important parts of oils that do not break down. They have a lot of health benefits, such as lowering cholesterol, reducing inflammation, eliminating bacteria and fungi, and treating hyperlipoproteinemia. Some people also think they can help lower blood sugar and glycated hemoglobin levels, raise insulin levels in the blood, and lower the risk of cancer (Zhang et al., 2020). Stigmasterol is a vital component in the production of semi-synthetic progesterone, a hormone that aids tissue recovery and influences estrogen function. Stigmasterol is also used to create estrogens, androgens, corticoids, and vitamin D3. In the past, the Upjohn Company employed stigmasterol to make cortisone (Doan et al., 2019). Beta-sitosterol, campesterol, and stigmasterol are all types of phytosterols that are found in many seed oils. These chemicals are renowned for their bioactive properties, specifically how they protect cells from damage and help people with diabetes. There are a lot of diverse biological impacts that sterols in seed oils have. They have an effect on oxidative stress, inflammation, insulin sensitivity, and lipid metabolism.
 |
Fig. 3 Chromatogram of sterol |
The sterols content of C. rheedei seed oils.
3.4 Amino acids
There has not been any published research on the amino acid composition of C. rheedei seeds yet. The entire C. rheedei plant has 4334.413 mg/100 g of amino acids, with 12 different types. Table 4 shows what amino acids are in the seeds of C. rheedei. About 22.3% of the protein was made up of essential amino acids (EAA), which are regarded as markers of good proteins. This percentage is almost the same as the percentage of whole-wheat flour (around 27%) (Nuanmano et al., 2015), which is one of the most common sources of vegetable protein. The EAA to non-EAA ratio of olive seed proteins (0.86) is much greater than the FAO’s (1985) advised value of 0.38 for adult humans. The complete seeds of C. rheedei have satisfactory levels of six important amino acids: histidine, lysine, phenylalanine, threonine, and valine. Furthermore, glycine, proline, hydroxyproline, and proline are conditionally necessary non-essential amino acids. This means that the body can make them when healthy but not when sick. Hydroxyproline, glutamine, lysine, and serine (51.206–133.176 mg/100 g) are among the amino acids exceeding 50 mg/g. Proline is a crucial non-essential amino acid that supports the immune system and facilitates tissue healing (133 mg/g). Proline is abundantly present in beef, soy protein, cottage cheese, cream cheese, gelatine products, and cabbage (Nuanmano et al., 2015). Proline and hydroxyproline are essential for maintaining collagen stability, constituting around 23% of the amino acids in collagen molecules (Xu et al., 2019; Kumar et al., 2016). Serine and alanine are two amino acids that can soothe the skin. They alleviate burning and pain, reduce redness and irritation, and enhance skin comfort. These findings promise the creation of skincare products derived from C. rheedei. Consuming C. rheedei consistently may enhance your health due to its high protein content and essential amino acids and minerals necessary for bodily function. This work is the first investigation on the amino acids.
 |
Fig. 4 Chromatogram of amino acids |
The amino acid compositions in C. rheedei seeds.
3.5 Antioxidant activity
The results of the study demonstrate that C. rheedei seed oil is a highly potent antioxidant. The DPPH technique (IC50 = 13.05 ± 0.96 μg/mL), FRAP (2855.53 μmol Fe2+/L), and ABTS (EC50 = 22.78 ± 3.99 μg/mL) all show this. The measurement is very accurate, as shown by the correlation coefficient (r2 ≥ 0.99). Zanzabil et al. (2023) conducted a study demonstrating that the roots of C. rheedei effectively scavenge nitric oxide radicals, exhibiting an IC50 of 28.3 μg/mL, which surpasses the efficacy of ascorbic acid, which has an IC50 of 18.9 μg/mL (Mahmud et al., 2019). We observed that the aqueous and ethanol extracts of C. rheedei at concentrations of 25, 50, 75, 100, and 250 μg/mL indicated that the ethanol extract exhibited superior advantages at lower doses (25 μg/mL). This means that the solvent used to extract the antioxidant effect has an effect. These results are lower than the control, but they nonetheless show that C. rheedei seed oil has a lot of antioxidant potential. This is very different from olive oil, which is known for its innate ability to fight free radicals. The distinction is based on the kind and how it was created.
Benavente-García et al. (2000) and Bendini et al. (2007) assert that the IC50 DPPH of extra virgin olive oil typically ranges from 20 to 80 μg/mL, contingent upon its polyphenol and tocopherol content (Zanzabil et al., 2019; Benavente–García et al., 2000). The FRAP value for pure olive oil is usually between 1500 and 2500 μmol Fe2+/L, whereas the ABTS-TEAC value for pure olive oil is usually between 1000 and 2000 μmol Trolox/kg. C. rheedei seed oil has a lower IC50 value than olive oil, which means it is better at getting rid of free radicals in vitro. The FRAP value of C. rheedei oil (2855.53 μmol Fe2+/L) is also higher than the average for olive oil. This means it can break things down more, which could be because it has α-tocopherol (21.13 mg/100 g) and γ-tocopherol (9.26 mg/100 g) and phytosterols like β-sitosterol and stigmasterol. C. rheedei oil has a lot of oleic acid (71.38%), which is similar to olive oil. This indicates that it can help with both anti-inflammatory and anti-diabetic effects, which are two things that long-term oxidative stress can induce. But olive oil is better because it has polyphenols like oleuropein and hydroxytyrosol, which are powerful compounds that protect cells and DNA from damage caused by oxygen. More studies are needed to find out exactly what polyphenols are in C. rheedei oil.
3.6 Antidiabetic activity
The observed in vitro α-glucosidase inhibition may be attributed in part to oleic acid, tocopherols, and phytosterols. However, the exact mechanisms remain unclear and require further study. Our discussion is limited to in vitro inhibitory activity.
The inhibitory effect of seed oil (hexane extract residue) on α-glucosidase has been evaluated and compared with acarbose, a standard antidiabetic drug. As shown in Table 6, C. rheedei seed oil exhibits a strong inhibitory effect with an IC50 value of 102.4 ± 3.86 μg/mL, demonstrating a stronger α-glucosidase inhibitory capacity than acarbose (198.5 ± 6.25 μg/mL). Previous studies continue to support the anti-diabetic potential of C. rheedei (Ramjith et al. 2013) reported that the aqueous leaf extract significantly reduced fasting blood sugar levels in diabetic mice (Bendini et al., 2007). At doses of 200 mg/kg and 400 mg/kg, the extract showed IC50 values of 135.5 ± 3.21 and 123.66 ± 5.73 μg/mL, respectively, with effects comparable to the standard drug Glibenclamide (5 mg/kg, IC50 = 131 ± 3.48 μg/mL).
The comparison between C. rheedei seed oil and olive oil reveals a notable difference. In a study by Jacinta G et al. (2017), the extract from pure olive oil had an IC50 value for α-glucosidase of 152.8 μg/mL, lower than that of C. rheedei. This difference may stem from the high presence of oleic acid, tocopherol, and phytosterol in C. rheedei seed oil, compounds that play an active role in improving insulin sensitivity and regulating blood sugar levels. Considering the ability to inhibit the α-glucosidase enzyme, C. rheedei seed oil exhibits superior activity compared to pure olive oil under in vitro conditions, which points to the potential application of this seed oil as a natural diabetes treatment agent. In addition, traditional uses suggest no acute toxicity, supporting the preliminary food safety of these seeds. Given their high oil content and bioactivity, C. rheedei seeds also hold promising economic potential for nutraceutical and functional food applications in Southeast Asia.
The in vitro bioactivity of seed oil.
 |
Fig. 5 (A) C. rheedei seeds in Vietnam. (B) C. rheedei seed oil extract in Vietnam. |
4 Conclusions
This study provides the first comprehensive nutritional and functional profile of C. rheedei (C. rheedei J.F. Gmelin) seeds and their oil. The seeds yielded 32.6% oil, dominated by oleic acid (71.4%) and containing palmitic, linoleic, and linolenic acids in a balanced ω-3/ω-6 ratio (∼1:1). The high oleic acid content supports cardiovascular and glycemic health while also contributing moisturizing and barrier-protective properties relevant for cosmetic applications. Tocopherols (mainly α- and γ-isomers) and phytosterols (β-sitosterol and stigmasterol) were abundant, conferring antioxidant and anti-inflammatory potential.
For the first time, the amino acid profile of C. rheedei seeds was reported. Essential amino acids accounted for 22.3% of the total, while non-essential amino acids such as proline and hydroxyproline contribute to collagen stability and tissue repair, and serine and alanine may impart skin-calming effects.
Functionally, the seed oil showed strong antioxidant activity (DPPH IC50 = 13.1 μg/mL; FRAP = 2855.5 μmol Fe2+/L; TEAC EC50 = 22.8 μg/mL) and significant in vitro α-glucosidase inhibitory activity (IC50 = 102.4 μg/mL), surpassing the standard drug acarbose (198.5 μg/mL). These findings confirm the novelty of this work and indicate that C. rheedei seeds are a promising new resource for functional food and nutraceutical development.
Preliminary toxicological studies suggest no acute or subacute toxicity of C. rheedei extracts, but comprehensive safety and allergenicity assessments remain necessary. With its relatively high oil yield and bioactive composition, C. rheedei offers economic potential for cultivation and processing in Southeast Asia, with applications extending to functional foods, nutraceuticals, and cosmetics. Future research should focus on LC–MS-based profiling of minor phytochemicals and in vivo validation and explore more extraction methods or varieties of the C. rheedei for agronomic optimization and industrial production to support the sustainable utilization of this underexplored species.
Acknowledgments
We would like to thank the mission: “Support scientific research activities for senior researchers in 2024–2025”, code: NVCC 07.04 /24-25 and the technical support provided by the members of the Working Group of Lipid Research at the Department for Safety and Quality for Cereals in Detmold, Germany of the Max-Rubner-Institut. for supporting this study.
Conflicts of interest
The authors declare that they hold no competing interests.
References
- Alcubierre N, Navarrete-Munoz EM, Rubinat E, et al. 2016. Association of low oleic acid intake with diabetic retinopathy in type 2 diabetic patients: a case–control study. Nutr. Metab. 13(1): 40. https://doi.org/10.1186/s12986-016-0099-5. [Google Scholar]
- Benavente-García O, Castillo J, Lorente J, Ortuno A, Del Rio JA. 2000. Antioxidant activity of phenolics extracted from Olea europaea L. leaves. Food Chem. 68: 457–462. [Google Scholar]
- Bendini A, Cerretani L, Carrasco-Pancorbo A, et al. 2007. Phenolic molecules in virgin olive oils: a survey of their sensory properties, health effects, antioxidant activity and analytical methods. An overview of the last decade. Molecules 12(8): 1679–719. https://doi.org/10.3390/12081679. [CrossRef] [PubMed] [Google Scholar]
- Bykhovets M. 2010. Studying fatty acid content in red blood cell membranes in diabetic retinopathy of type 2 diabetes patients. J Educ. Health Sport 10(10): 51–60. https://doi.org/10.12775/JEHS.2020.10.10.005. [Google Scholar]
- Chen H, Yan X, Lin W, Zheng L, Zhang W. 2004. A new method for screening α-glucosidase inhibitors and application to marine microorganisms. Pharm. Biol. 42(6): 416–421. https://doi.org/10.1080/1388020049088598 [Google Scholar]
- Doan LP, Nguyen TT, Pham MQ, Tran QT, Pham QL, Tran DQ et al. 2019. Extraction process, identification of fatty acids, tocopherols, sterols and phenolic constituents, and antioxidant evaluation of seed oils from five Fabaceae species. Processes 7(7): 456. https://doi.org/10.3390/pr7070456. [Google Scholar]
- Etienne Guillocheau, Philippe Legrand, Vincent Rioux. 2017. Benefits of natural dietary trans fatty acids towards inflammation, obesity and type 2 diabetes: defining the n-7 trans fatty acid family. Oils Fats Crops Lipids, 26, 46. https://doi.org/10.1051/ocl/2019047. [Google Scholar]
- García-inza GP, Castro DN, Hall AJ, Rousseaux MC. 2016. Opposite oleic acid responses to temperature in oils from the seed and mesocarp of the olive fruit. Eur. J. Agron. 76: 138–147. DOI:10.1016/j.eja.2016.03.003. [Google Scholar]
- Gomez – Caravaca AM, Verardo V, Caboni MF. 2010. Chromatographic techniques for the determination of alkyl-phenols, tocopherols and other minor polar compounds in raw and roasted cold pressed cashew nut oils. J.f Chromatogr. A 1217(47): 77411–74117. DOI: https://doi.org/10.1016/j.chroma.2010.09.054. [Google Scholar]
- Gunstone FD, Harwood JL. Occurrence and characteristics of oils and fats, In: The Lipid Handbook with CD-ROM, 3rd ed... Boca Raton: CRC Press, 2007, 37–141. https://doi.org/10.1201/9781420009675.ch2. [Google Scholar]
- Hakamata W, Kurihara M, Okuda H, Nishio T, Oku T. 2016. Design and screening strategies for α-glucosidase inhibitors based on enzymological information. Curr. Top. Med. Chem. 9(1): 3–12. https://doi.org/10.2174/156802609787354306 [Google Scholar]
- Hiepko P, Opiliaceae I, Anderson CE, Marhold K. 2008. Species Plantarum: Flora of the World. Conservatoire et Jardin botaniques de la Ville de Genève 12: 1–71. [Google Scholar]
- International Organization for Standardization. 1988. Animal and vegetable fats and oils-Gas chromatography of fatty acid methyl esters, Part 2: Preparation of methyl esters of fatty acids, 2nd Edn., ISO, Geneva, Switzerland, Standard No. 5509. [Google Scholar]
- International Organization for Standardization. 2016. Animal and vegetable fats and oils - Determination of tocopherol and tocotrienol contents by high-performance liquid chromatography, 3rd Edn., ISO, Geneva, Switzerland, Standard No. 9936. (ISO 9936:2016). [Google Scholar]
- International Olive Council. 2001. Determination of the composition and content of sterols by capillary-column gas chromatography. Spain; Madrid, COI/T, 20/Doc. No. 10 Rev. [Google Scholar]
- International Olive Council, Determination of the composition and content of sterols and triterpene dialcohols by capillary column gas chromatography. Spain; Madrid, COI/T, 20/Doc. No. 30/Rev, 2013. [Google Scholar]
- ISO 659. 2009. Oilseeds – Determination of oil content (Reference method). EN ISO 659. [Google Scholar] [Google Scholar]
- Jacinta G, Clara G, Patricia V, et al. 2017. Inhibition of α-glucosidase and α-amylase by Spanish extra virgin olive oils: the involvement of bioactive compounds other than oleuropein and hydroxytyrosol, Food Chem. 235(15): 298–307. [Google Scholar]
- Jaudzems G, Guthrie J, Lahrichi S, Fuerer C. 2018. Total Amino acids by UHPLC-UV in Infant Formulas and Adult Nutritionals, J. AOAC Int. 102(5): 1574–1588. https://doi.org/10.1093/jaoac/102.5.1574. [Google Scholar]
- Kumar S, Khare P, Kumar NH, Raghuwanshi N, Srivastava R. 2016. Hydroxyproline: a potential biochemical marker and its role in the pathogenesis of different diseases. Curr. Protein Pept. Sci. 17(6): 596–602. https://doi.org/10.2174/1389203717666151201192247 [Google Scholar]
- Maestri D, Barrionuevo D, Bodoira R, Zafra A, Jiménez-López J, Alché JDD. 2019. Nutritional profile and nutraceutical components of olive (Olea europaea L.) seeds. J Food Sci. Technol. 56(9): 4359–4370. doi: 10.1007/s13197-019-03904-5. [Google Scholar]
- Mahmud ZA, Bachar SC, Hasan CM, Emran TB, Qais N, Uddin MMN. 2017. Phytochemical investigations and antioxidant potential of roots of Leea macrophylla (Roxb.). BMC Res. Notes 10: 1–9. https://doi.org/10.1186/s13104-017-2503-2. [Google Scholar]
- Massimo C, Lucio T, Jesus MA, Giovanni L, Caramia GM. 2009. Extra virgin olive oil and oleic acid. Nutr. Clin. Diet. Hosp. 29(3): 12–24. [Google Scholar]
- Mounnissamy V.M., Kavimani S., Sankari G., Quine S.D. and Subramani K., 2010. Evaluation of acute and sub-acute toxicity of ethanol extracts of i J.Gmelin (Opiliaceae). J. Brew. Distill. 1(1): 011–014. [Google Scholar]
- Mostafa O, Mohamed O, Awatif I, Mohamed AS, Bertrand M. 2021. Characterisation of different parts from Moringa oleifera regarding protein, lipid composition and extractable phenolic compounds. Oils Fats Crops Lipids, 28, 45. https://doi.org/10.1051/ocl/20210352021. [Google Scholar]
- Mounnissamy VM, Subramanian KSK, Gnanapragasam SGS, Dhayalamurthi S, Quine SD, Subramani K. 2009. Effect of i C. rheedei C. rheedei J.F. Gmelin (Opiliaceae) on diuretic activity in rats. J. Pharm. Res. 2(10): 1627–1628. http://jpronline.info/article/view/760/630. [Google Scholar]
- Nuanmano S, Prodpran T, Benjakul S. 2015. Potential use of gelatin hydrolysate as plasticizer in fish myofibrillar protein film. Food Hydrocoll. 47: 61–68. https://doi.org/10.1016/j.foodhyd.2015. 01.005. [Google Scholar]
- Özcan MM, Juhaimi FAL, Uslu N, Ghafoor K, Ahmed IAM, Babiker EE. 2019. The effect of olive varieties on fatty acid composition and tocopherol contents of cold pressed virgin olive oils. J. Oleo Sci. 68(4): 307–310. https://doi.org/10.5650/jos.ess18251. [Google Scholar]
- Ramjith US, Roopitha P, Cyril Mathew Jacob. 2013. Isolation anti-diabetic and antioxidant evaluation of aqueous extract of Cansjera rheedii leaves. Asian J Pharm. Clin. Res. 6(3): 228–234. [Google Scholar]
- Saeed N, Khan RM, Shabbir M. 2012. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement. Altern. Med. 12: 221. https://doi.org/10.1186/1472-6882-12-221. [Google Scholar]
- Salehi B, Ata A, Anil-kumar NV, et al. 2019. Antidiabetic potential of medicinal plants and their active components. Biomolecules 9(10): 551. https://doi.org/10.3390/biom9100551. [Google Scholar]
- Salazar C, Armenta JM, Shulaev V. 2012. An UPLC-ESI-MS/MS assay using 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate derivatization for targeted amino acid analysis: application to screening of Arabidopsis thaliana mutants. Metabolites 2(3): 398–428. https://doi.org/10.3390/metabo2030398. [Google Scholar]
- Simopoulos AP. 1996. The role of fatty acids in gene expression: health implications. Ann.f Nutr. Metabol. 40: 303–311. https://doi.org/10.1159/000177929. [Google Scholar]
- Soumya D, Srilatha B. 2011. Late stage complications of diabetes and insulin resistance. J. Diabetes Metab. 2(9): 1000167. https://doi.org/10.4172/2155-6156.1000167. [Google Scholar]
- Stephen I, Revathi G, Pradeepha P, Arthy M, Swathi PR, Mounnissamy VM. 2018. Diuretic activity of rutin isolated from J. Gmelin (Opiliaceae). World J. Pharm. Med. Res. 4(5): 260–262. [Google Scholar]
- Teo ZL, Tham YC, Yu M, Chee ML, Rim TH, Cheung N et al. 2021. Global prevalence of diabetic retinopathy and projection of burden through 2045: systematic review and meta-analysis. Ophthalmology 128(11): 1580–1591. https://doi.org/10.1016/j.ophtha.2021.04.027. [Google Scholar]
- Wanna K, Wipada K. 2007. Determination of Some Fatty Acids in Local Plant Seeds. Chiang Mai J. Sci. 34(2): 249–252. [Google Scholar]
- Worachai W, Supawan T, Akkasit J. 2023. Effects of aqueous extraction factors on commercial protease inhibitory activities, phenolic acid profiles and chemical properties of Hom Mali-105 rice (Oryza sativa L.) bran extract. Chiang Mai J. Sci. 50(5): 1–14. https://doi.org/10.12982/CMJS.2023.054. [Google Scholar]
- Wunderle C, Haller L, Laager R, et al. 2024. The association of the essential amino acids lysine, methionine, and threonine with clinical outcomes in patients at nutritional risk: secondary analysis of a randomized clinical trial. Nutrients 16(16): 2608. https://doi.org/10.3390/nu16162608. [Google Scholar]
- Xu S, Gu M, Wu K, Li G. 2019. Unraveling the role of Hydroxyproline in maintaining the thermal stability of the collagen triple helix structure using simulation. The J Phys. Chem. B 123(36): 7754–7763. https://doi.org/10.1021/acs.jpcb.9b05006. [Google Scholar]
- Yuvaraj P, Subramoniam A, Louis T, Madhavachandran V, Narasu ML. 2013. Attenuation of expression of cytokines, oxidative stress and inflammation by hepatoprotective phenolic acids from Thespesia populnea Soland ex Correa stem bark, Ann. Phytomed. 2(2): 47–56 [Google Scholar]
- Zhang T, Xie L, Liu R, et al. 2020. Revisiting the 4,4‐dimethylsterols profile from different kinds of vegetable oils by using GC‐MS. LWT–Food Sci. Technol. 124: 109163. https://doi.org/10.1016/J.LWT.2020.109163. [Google Scholar]
- Zanzabil KZ, Hossain MS, Hasan MK. 2023. Diabetes mellitus management: an extensive review of 37 medicinal plants. Diabetology 4(2): 186–234. https://doi.org/10.3390/diabetology4020019. [Google Scholar]
Cite this article as: Pham Q.L, Nguyen T.N, Tuyet Dang T.M, Dang T.P.L, Matthäus B, Nguyen L.N, Dinh T.H, Duong T.T, Doan L.P. 2026. The fatty acid and amino profiles, bioactive compounds, and alpha glucosidase inhibition activity of C. rheedei J.F. Gmelin seed oil . OCL 33: 15. https://doi.org/10.1051/ocl/2026005
All Tables
All Figures
 |
Fig. 1 Chromatogam of fatty acids |
| In the text | |
 |
Fig. 2 Chromatography of tocopherol |
| In the text | |
 |
Fig. 3 Chromatogram of sterol |
| In the text | |
 |
Fig. 4 Chromatogram of amino acids |
| In the text | |
 |
Fig. 5 (A) C. rheedei seeds in Vietnam. (B) C. rheedei seed oil extract in Vietnam. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.




