| Issue |
OCL
Volume 33, 2026
|
|
|---|---|---|
| Article Number | 12 | |
| Number of page(s) | 21 | |
| Section | Agronomy | |
| DOI | https://doi.org/10.1051/ocl/2025043 | |
| Published online | 17 March 2026 | |
Review
To shatter or not to shatter: synthesizing biology and engineering for mechanised harvesting of sesame
Éclater ou ne pas éclater: synthèse des approches biologiques et d’ingénierie pour la récolte mécanisée du sésame
1
Seed and Plant Improvement Institute, Agricultural Research, Education and Extension Organisation (AREEO), Karaj, Iran
2
Agricultural Technical and Engineering Research Department, Golestan Agricultural and Natural Resources Research and Education Center, Agricultural Research, Education and Extension Organization (AREEO), Gorgan, Iran
3
Department of Genetics and Plant Breeding, Faculty of Agriculture and Natural Resources, Imam Khomeini International University, Qazvin, Iran
4
School of Biological Sciences,The University of Western Australia, Crawley, 6009, WA 6009, Australia
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
9
August
2025
Accepted:
19
December
2025
Abstract
Seed shattering remains a major barrier to mechanised harvesting of sesame (Sesamum indicum L.). It reduces yield potential, with reported losses that can exceed 50%, and hinders farmers’ adoption of modern harvesting technologies. This review bridges biological and engineering disciplines by providing an integrative analysis of the genetic and morphological factors in seed retention, alongside the agronomic and mechanical strategies required for efficient harvesting. Key capsule traits, such as dehiscence angle, membrane development, and seed-placental adhesion, are examined for their roles in seed retention and suitability for mechanised harvesting. Quantitative methods for assessing shattering intensity, including the TI/KE/TO index (a score for upright retention, inverted retention, and tip opening) and Langham’s capsule breakage protocol, are discussed. The review further explores advances in breeding, including the utilisation of landraces and exotic germplasm, marker-assisted selection, and identification of candidate genes and QTLs linked to shattering resistance. Mechanised harvesting methods, particularly the choice between direct and two-stage approaches, are evaluated under varying environmental conditions, with an emphasis on seed quality preservation and economic efficiency. Finally, this review is the first to propose a comprehensive ideotype framework that resolves the fundamental trade-off between field-level seed retention and mechanical threshability by systematically integrating key biological traits with engineering parameters
Résumé
La déhiscence des capsules demeure un obstacle majeur à la récolte mécanisée du sésame (Sesamum indicum L.). Elle réduit fortement le rendement final, avec des pertes pouvant dépasser 50%, et freine l’adoption des technologies modernes de récolte par les agriculteurs. Cette revue fait le lien entre biologie et ingénierie en proposant une analyse intégrative des facteurs génétiques et morphologiques impliqués dans la rétention intra-capsulaire des graines, ainsi que des stratégies agronomiques et mécaniques nécessaires à une récolte efficace. Les principaux traits des capsules – tels que l’angle d’ouverture, le développement des membranes et l’adhérence de la graine – sont examinés pour leur rôle dans la rétention des graines et leur aptitude à la récolte mécanisée. Les méthodes quantitatives d’évaluation de l’intensité de déhiscence, notamment l’indice TI/KE/TO (score de rétention en position verticale, rétention en position inversée et ouverture de l’extrémité) ainsi que le protocole de rupture de capsule de Langham, sont discutées. La revue explore également les avancées en amélioration génétique, incluant l’utilisation de variétés locales et de ressources exotiques, la sélection assistée par marqueurs et l’identification des gènes candidats et des loci de caractères quantitatifs (QTL) associés à la résistance à la déhiscence. Les méthodes de récolte mécanisée, en particulier le choix entre une approche directe ou en deux étapes, sont évaluées selon différents contextes environnementaux, avec un accent mis sur la préservation de la qualité des graines et l’efficacité économique. Enfin, cette revue est la première à proposer un cadre idéotypique complet permettant de résoudre le compromis fondamental entre la rétention des graines au champ et l’adaptation au battage mécanique, en intégrant de façon systématique les principaux traits biologiques et les paramètres d’ingénierie.
Key words: Capsule shattering / capsule traits / mechanized harvesting / seed retention traits / Sesamum indicum
Mots clés : Déhiscence des capsules / traits de capsules / récolte mécanisée / rétention des graines / Sesamum indicum
© M. Gholamhoseini et al., Published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Highlights
Integrates biological and engineering approaches to address sesame harvest mechanization.
Proposes a comprehensive ideotype framework balancing shatter resistance and threshability.
Defines the critical trade-off between seed retention in the field and mechanical release.
Links key capsule traits directly to optimal combine harvester settings and adjustments.
Reviews and contextualizes quantitative methods for assessing sesame shatter resistance potential.
1 Introduction
Sesame is a strategic oilseed crop in arid and semi-arid regions, playing a critical role in food security, rural livelihoods, and climate-resilient agriculture (Hamedani et al., 2022; Pathak et al., 2014). Also one of the oldest domesticated oilseeds, it remains highly valued globally. Its seeds typically contain over 50% oil, characterized by a high proportion of unsaturated fatty acids (primarily oleic and linoleic acids, often >80% combined) compared to saturated fats, contributing to its reputation for cardiovascular health benefits (Gholamhoseini, 2022a). The seeds are also rich in proteins and antioxidant lignans (Ashri, 2006; Andargie et al., 2021). Sesame is particularly well-suited to low-input, drought-prone farming systems, making it an essential option for sustainable agriculture in marginal environments (Dossa et al., 2017a).
Despite its advantages, sesame cultivation is constrained by significant challenges, foremost among them seed shattering. The reliance on manual harvesting–a direct consequence of shattering-creates a major production bottleneck, especially as the global sesame market is projected to grow annually by nearly 5.8% (Grand View Research, 2024). Global sesame production currently exceeds 6.5 million tons annually (FAOSTAT, accessed October 25, 2025), reflecting its significant economic role. This makes shattering not only an agronomic threat through direct yield loss but also a source of substantial socioeconomic burdens, including inflated labor costs, increased farmer risk, and barriers to profitable, large-scale mechanization.
This genetically regulated dehiscence process causes substantial pre-harvest and harvest-time yield losses (Uzun et al., 2002; Gholamhoseini et al., 2023a). Although capsule dehiscence and seed dispersal represent beneficial natural mechanisms for seed spread, this trait poses a significant barrier to the expansion of sesame farming. As illustrated in Table 1, the largest yield losses often occur during manual harvesting of traditional, shattering-prone varieties. Under typical practices (i.e., attempting timely harvest with standard manual methods), these operational harvest losses alone can range from approximately 6% to 15% of the potential yield, depending on the farming system and specific conditions (Usman et al., 2022). Furthermore, in scenarios involving significant harvest delays or inexperienced handling, particularly with these highly susceptible landraces, total losses (including pre-harvest shattering and operational losses) can easily exceed 50%, highlighting the crop’s inherent vulnerability (Gholamhoseini and Dolatabadian, 2024a).
These losses are aggravated by the crop’s indeterminate growth habit, asynchronous capsule maturity, and dependence on labour-intensive harvesting practices. Mechanical harvesting remains rare in many sesame-producing countries, partly due to the fragility of traditional landraces and the lack of compatible agronomic traits, such as uniform maturity, non-shattering capsules, and suitable plant architecture (Langham and Wiemers, 2002).
Nevertheless, the successful mechanisation programs in several key countries offer promising insights. In countries such as Brazil, the USA, and China, the combination of shatter-resistant cultivars and adapted mechanisation technologies has transformed sesame into a commercially viable and scalable crop. For instance, Brazil exemplifies this transformation. The country’s sesame cultivated area has surged dramatically in recent years, with production reaching approximately 361000 tons in the 2023/24 season, a significant increase driven by varietal improvement and widespread mechanization (FAOSTAT, accessed October 25, 2025). This rapid expansion has positioned Brazil among the world’s top sesame exporters, demonstrating how integrating technology and breeding can unlock production potential on a global scale.
To address these challenges, this review synthesizes anatomical, physiological, and engineering insights to propose a sesame ideotype compatible with mechanised harvesting. While past reviews have often addressed either the genetics of shattering or harvesting technologies in isolation, this paper fills a critical gap by providing an integrated analysis that bridges these two fields. The integration of biological resistance and harvesting innovations holds the key to enhancing sesame productivity, economic viability, and global competitiveness, especially in regions where sesame remains an underutilised yet high-potential crop.
Estimated sesame yield losses along the production-to-consumption chain.
2 Capsule anatomy and mechanism of seed shattering in sesame
A comprehensive understanding of capsule anatomy is essential for designing both effective breeding strategies and mechanised harvesting technologies to reduce seed shattering. In sesame, the fruit is a capsule and its anatomical and histological structure plays a critical role in its natural tendency to shatter upon maturation (Bedigian, 2010). The sesame capsule is composed of three primary tissue layers: the epicarp (outer layer), the mesocarp (middle layer), and the endocarp (inner layer) (Day, 2000a). Each of these layers contributes differently to the biomechanics of seed retention or release.
The mesocarp consists of soft parenchymatous cells, while the endocarp is composed of heavily lignified sclerenchymatous cells, contributing rigidity and structural support (Day, 2000b). These endocarp cells form a hardened sheath around the locules but do not extend toward the placenta. Seeds are attached centrally within the capsule via placental connections (Fig. 1).
As the capsule matures and dries, longitudinal dehiscence occurs, starting from the apex and proceeding downward. This dehiscence is similar to that observed in other dry fruits, such as siliques in Brassica species. Two primary structural prerequisites have been identified for fruit dehiscence (Maity et al., 2021):
Differential thickening or cell wall orientation among adjacent tissue layers, and
The presence of a predetermined zone of weakness, or abscission layer, along the capsule.
In sesame, the endocarp shrinks less than the mesocarp during dehydration. This disparity creates internal mechanical stress, promoting delamination between the layers and facilitating capsule opening. The greater the differential shrinkage, the higher the tendency for the capsule to split. Moreover, the orientation of sclerenchymatous fibres in the endocarp is not uniform along the capsule’s longitudinal axis (Shtein et al., 2016a), which contributes to both lateral dehiscence along the false septum and splitting from the capsule tip.
A critical feature in this process is the zone of weakness located in the false septum, a membranous structure separating the locules. This septum includes thin-walled parenchymatous cells that lie between vascular bundles and the epicarp. During early capsule development, partial enzymatic degradation of the parenchymatous cell walls occurs, primarily involving cellulases and pectinases, further weakening this region (Day, 2000c). Collectively, the asynchronous shrinkage of mesocarp and endocarp tissues, combined with the formation of the false septum as a structural weak point, creates the biomechanical basis for capsule dehiscence and seed shattering in sesame.
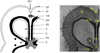 |
Fig. 1 Cross-section of a sesame capsule. Photo from Day (2000a). SU: Sutural gap, CS: Carpel wall, EN: Endocarp, EP: Epicarp, FS: False septum (pseudoseptum), L: Lumen, M: Mesocarp, P: Placenta, S: Seed, VB: Vascular bundle of the central axis. |
2.1 Capsule Traits Contributing to Seed Retention in Sesame
A standardised set of capsule traits is used to assess sesame shattering resistance, optimising both field seed retention and mechanised harvesting efficiency (Langham, 2011; Miao et al., 2021). These morphological and anatomical indicators guide breeders in identifying genotypes suited to mechanical systems. The key traits, their scoring methods, and optimal ranges for mechanised harvesting are detailed in Table 2. These morphological indicators assess different aspects of both external and internal capsule integrity. Key traits include the extent of the capsule’s longitudinal split (capsule dehiscence score) and the degree of opening at its apex (capsule valve opening score), which influences drying and initial seed loss. Internally, crucial traits are the completeness of the membrane covering the seed cavity (membrane coverage score), the overall tightness of the dried capsule (capsule compactness score), the strength of the seed’s attachment to the central placenta (seed–placenta adhesion score), and the proximity of the membrane to the placental axis (membrane-placenta contact score) (Fig. 2).
Two qualitative traits further enhance retention: a non-curved capsule tip (Fig. 3) reduces rupture risk, and an upright capsule angle improves stability by minimising shake-induced dehiscence. Together with the quantified traits, these features strongly influence a genotype’s suitability for mechanised harvest and resistance to preharvest shattering (Langham and Wiemers, 2002; Queiroga et al., 2019). However, a major limitation in utilizing these internal traits (e.g., membrane coverage) is the reliance on destructive and labor-intensive manual scoring. Unlike external features, assessing internal capsule architecture remains a bottleneck for high-throughput screening, highlighting the need for future research into non-destructive imaging techniques.
 |
Fig. 2 Key morphological capsule traits influencing sesame seed retention and threshability. (a) Capsule dehiscence score: compares a generally shatter-resistant genotype (left) exhibiting a more complete suture split (score closer to 1), which facilitates seed release during threshing, versus a susceptible genotype (right) with a less extensive split. (b) Capsule valve opening score: illustrates differences in apical gap width (optimal score ∼ 7 allows drying while retaining seeds). (c) Membrane coverage score: shows optimal coverage (score 7), which retains seeds pre-harvest but is fragile enough for threshing. (d) Capsule compactness (cross-section): highlights reduced apical gap in resistant (left) vs. susceptible (right) capsules (optimal score 4-5 balances retention and release). (e) Seed-placenta adhesion: illustrates seed attachment within the locules (optimal score ∼ 7 withstands vibration but yields to threshing). Refer to Table 2 for detailed scoring. Scale bar in (a) = 1 cm |
Summary of key morphological capsule traits for assessing shatter resistance.
 |
Fig. 3 Capsule A has tip curvature after drying, making it unsuitable for mechanical harvesting; Capsule B lacks curvature but lacks shattering resistance. Capsule C combines a non-curved tip with shattering-resistant traits, making it optimal. Photo by the authors. |
2.2 Quantification of seed shattering intensity
Reliable, standardised methods are crucial for evaluating seed retention and breeding shatter-resistant sesame. Two main systems are used: the TI/KE/TO Index (a numerical score for upright retention, inverted retention, and tip opening of capsules), developed by the US-based company Sesaco (Sesame Coordinators), and the Langham Method (Langham, 2013).
2.2.1 The TI/KE/TO index: a numerical descriptor of shattering resistance
The TI/KE/TO Index is a three-digit score (0−8 for each digit) evaluating capsule seed retention under different conditions:
TI (Upright Retention): This parameter assesses the seeds retained in capsules that have been allowed to dry naturally on the plant under field conditions. These vertically oriented capsules are then scored from 0 (empty) to 8 (full).
KE (Inverted Retention): This score quantifies the capsule’s ability to retain seeds when it is inverted and gently shaken, simulating the vibrational forces experienced prior to and during combine harvesting. It is considered the most critical indicator of suitability for mechanisation. Scores range from 0, where all seeds fall out easily, to 8, where seeds remain firmly attached. An optimal KE score for mechanised systems is typically in the mid-range (e.g., 4 to 6), indicating a genotype that resists field shattering but allows for efficient seed release during threshing.
TO (Tip Opening): This measures the degree of separation between the capsule valves at the apex after drying, scored on a scale from 1 (fully dehisced or wide open) to 8 (completely closed). While a low score indicates high shattering risk, a score of 8 is also undesirable as a fully closed tip can trap moisture and impede threshing. An optimal TO score is typically around 7, where the tip is only slightly open, allowing the capsule to dry effectively while still providing strong seed retention.
To measure the TI/KE/TO index:
Select three mature, dried capsules from the mid-section of three plants.
Open each capsule longitudinally and score seed retention (TI scores).
Repeat with inverted capsules and record KE scores.
Assess the degree of tip opening and assign TO scores.
Average the scores across the nine capsules (three per plant × three plants) for each component.
Table 3 summarises the TI/KE/TO indices of several Iranian cultivars, while Table 4 provides threshold TI/KE/TO values recommended for various mechanical harvesting techniques.
To comprehensively assess seed retention stability, measurements should be conducted at four post-maturation stages: (1) immediately after physiological maturity and initial drying, (2) after two weeks of favorable weather, (3) following one week of unfavorable conditions (e.g., rain or high humidity), and (4) one month post-drying under continued adverse weather. This protocol distinguishes genotypes with short-term shattering resistance from those with durable seed retention under field conditions (Gholamhoseini et al., 2024a). Critically, while the TI/KE/TO index provides detailed morphological descriptors essential for breeding specific traits, it remains semi-quantitative and subject to scorer bias. Therefore, it is most effective when used as a preliminary selection tool, ideally complemented by direct physical retention assays for final validation.
TI/KE/TO Index for evaluating seed shattering resistance of Iranian sesame cultivars.
Relationship between the TI/KE/TO Index and the harvesting method.
2.2.2 Langham’s method: a physical seed retention assay
In contrast to the semi-quantitative TI/KE/TO index, Langham’s (2013) method provides a direct physical assay to quantify shattering resistance. The core principle of this protocol is to harvest capsules precisely at physiological maturity—a stage typically reached around 40-50 days after flowering (depending on genotype and environment) (Langham, 2007) and visually indicated when approximately 75% of seeds within the mid-stem capsules change colour and exhibit a distinct dark hilum or longitudinal line (as shown in Fig. 4)-allow them to air-dry under controlled conditions, and then calculate the percentage of seeds retained after the capsules are simply inverted. This approach yields a clear, quantitative metric of seed retention. A major contribution of this method was its establishment of a practical classification system, where the >75% retention threshold became a widely accepted benchmark for identifying genotypes as resistant and suitable for mechanisation (Langham, 2000; Sadeghi et al., 2022). While this assay is highly accurate for final selection in breeding programs, its manual nature may limit its application for high-throughput screening in early generations.
Capsules are classified into four shattering susceptibility categories based on seed retention: (1) Highly shattering (<25%; unsuitable for mechanical harvesting), (2) Shattering (2550%; still non-viable), (3) Moderately resistant (50-75%; marginal for commercial harvest), and (4) Resistant (>75%; optimal for mechanisation with minimal loss) (Langham, 2000, 2013). This framework uses the 75% threshold as the key benchmark for mechanisation potential (Sadeghi et al., 2022). Table 5 shows Langham test results for Iranian cultivars.
 |
Fig. 4 Mature sesame seeds exhibiting the visual cues of physiological maturity, including colour change and a distinct dark hilum or longitudinal line (indicated by arrows), the stage targeted for sampling in Langham’s shatter resistance assay. Photo by the authors. |
Langham’s index for evaluating seed shattering resistance in Iranian sesame cultivars.
2.3 Genetic and breeding approaches for enhancing seed retention in sesame
The increasing adoption of mechanised harvesting is fundamentally reshaping sesame production globally. This trend is exemplified by its standardisation in the United States (Langham and Wiemers, 2002) and the rapid expansion in Brazil (Queiroga et al., 2019), where the integration of mechanisation-ready cultivars has been a key driver in boosting national production. Consequently, breeding for stable genetic resistance to seed shattering is a top priority. Agronomic adjustments offer short-term relief, but lasting progress relies on genetic improvement (Qureshi et al., 2022; Gholamhoseini and Dolatabadian, 2024a). This section reviews strategies from traditional phenotypic selection to modern molecular approaches for developing machine-compatible sesame ideotypes.
2.3.1 Selection based on morphological indicators
Empirical observations across diverse environments have underscored the heritability of certain morphological traits associated with seed retention (Tinak Ekom et al., 2025). Key features include capsule dehiscence angle, development of inner membranes across locule cavities, firmness of seed adhesion to the placenta, and the extent of capsule contraction during drying (Phumichai et al., 2017). As discussed in previous sections, these traits can be systematically scored, creating a reliable foundation for phenotypic selection. Traditional breeding programs have effectively utilised such descriptors in recurrent selection cycles to enhance seed retention capacity gradually. A prime example is the breeding program in the United States, which systematically applied morphological scoring for capsule traits to develop the first commercially viable, shatter-resistant cultivars suitable for combine harvesting (Langham, 2013).
2.3.2 Incorporating resistance through landraces and exotic germplasm
Significant progress in developing shatter-resistant sesame varieties has indeed been achieved globally over recent decades, driven by targeted breeding efforts in various key regions. Screening of diverse germplasm has successfully identified valuable sources for shattering resistance. For instance, studies on Ethiopian landraces have identified accessions with moderate but stable seed retention (Gebremichael, 2017), which are valuable for improving environmental adaptability. A pivotal breakthrough in developing non-shattering sesame was the discovery of the recessive indehiscent (id) gene mutation in a Venezuelan landrace (Qureshi et al., 2022). This single gene became the foundation for most of the shatter-resistant cultivars developed in the United States by breeders like Langham, leading to commercial varieties with near-complete seed retention. Similarly, breeding programs in Iran have successfully developed and released shatter-resistant varieties such as Mohajer and Barekat (Siahpoosh et al., 2024), which combine this trait with high yield and desirable agronomic characteristics. These documented genetic resources have been instrumental in creating modern, mechanisation-ready sesame cultivars
2.3.3 Challenges in trait integration
A fundamental challenge in sesame breeding is to resolve the inherent conflict between two opposing selection goals: shatter resistance and threshability. The ideal capsule must be robust enough to prevent pre-harvest seed loss in the field, yet simultaneously fragile enough to release its seeds efficiently during mechanical threshing (Teklu et al., 2022; Razaghi, 2024a). While tightly closed capsules effectively reduce pre-harvest losses, they may hinder seed release during threshing, thereby increasing harvest inefficiencies (Bedigian, 2004; Razaghi, 2024b). Breeding must therefore aim for a middle ground: ensuring capsule integrity during field drying while enabling clean seed release under mechanical impact. This balance is especially challenging due to the polygenic nature of shatter resistance, involving both structural and physiological factors (Uzun and Çağırgan, 2006; Sadeghi et al., 2022). Capsule architectural traits, such as locule wall thickness and partially fused capsule apices, have shown moderate to high heritability, offering potential for progress via conventional breeding (Yol, 2017). Nonetheless, combining traits like breakable yet strong membranes with moderate seed adhesion calls for advanced strategies, including marker-assisted selection (MAS) where possible.
2.3.4 Prospects for marker-assisted and genomic selection
To date, the molecular basis of seed shattering resistance in sesame remains only partially elucidated, a knowledge gap attributable to several key challenges. Primarily, shattering resistance is a complex quantitative trait, often controlled by multiple genes (QTLs) with small to moderate effects, making its genetic architecture difficult to dissect (Wang et al., 2023). Furthermore, sesame has historically received less research focus compared to major staple crops, resulting in a slower development of comprehensive genomic tools and resources needed for functional gene studies (Gholamhoseini and Dolatabadian, 2024a). However, the landscape of sesame genomics has been transformed by recent technological advances, particularly the advent of high-throughput Next-Generation Sequencing (NGS) and the subsequent development of a high-quality reference genome (Weldemichael and Gebremedhn, 2023). These tools have opened promising avenues for identifying genetic markers associated with key structural traits (Wang et al., 2022; Dossa et al., 2017a). Preliminary QTL mapping and transcriptome studies have highlighted genes involved in cell wall lignification (gene SIN_1005755) (Xu et al., 2021), pectin methylesterase activity (Wang et al., 2018 and 2023), and cellulose biosynthesis (Dossa et al., 2018), each contributing to the mechanical resilience of capsule tissues. More specifically, several resistance-related QTLs have been mapped on chromosomes 3, 5, and 7, providing key targets for marker-assisted breeding (Wang et al., 2023). A particularly significant finding is the identification of SiHEC3, a candidate gene on chromosome 3, where a specific deletion in its promoter region has been strongly correlated with delayed capsule opening and enhanced seed retention (Ju et al., 2024)
The validation of these candidate genes and QTLs is a critical next step. Once confirmed, these markers can be deployed in marker-assisted breeding programs to facilitate the introgression of resistance alleles into elite cultivars, thereby accelerating genetic gain without compromising other vital agronomic traits such as yield, oil content, or drought tolerance (Ataei et al., 2017). Prioritizing the development of robust, cost-effective markers for these specific QTLs and particularly for the functional polymorphism in the SiHEC3 gene, therefore, represents the most immediate and high-impact strategy for implementing MAS to improve shatter resistance in elite sesame backgrounds (Li et al., 2023). Despite these promising identifications, a significant gap remains in the functional validation of these markers across diverse genetic backgrounds. Most currently identified QTLs are population-specific, which limits their broad application in global breeding programs. Consequently, the development of diagnostic, gene-based markers that are stable across different environments represents the next critical priority for molecular research.
2.3.5 Toward ideotypes for mechanised harvest
The primary goal in modern sesame breeding is to develop capsule ideotypes that combine two opposing traits: maximal seed retention during pre-harvest desiccation and rapid, complete seed release during mechanical threshing (Langham and Wiemers, 2002; Uzun and Cagirgan, 2006; Gholamhoseini et al., 2024b). Achieving this goal requires optimising several key traits, each involving a fundamental trade-off. The ideal balance integrates specific biomechanical and physiological targets, as summarised in Table 6.
This goal demands coordinated research integrating next-generation phenotyping (e.g., micro-CT valve analysis), biomechanical modelling of harvester-pod interactions, and molecular breeding targeting loci for lignin deposition (CCoAOMT) and abscission layer development (polygalacturonase regulators) (Fuller and Allaby, 2009; Dong and Wang, 2015; Li and Olsen, 2016), validated through multi-environment field trials to match genotype, management, and machinery.
Key ideotype traits for mechanised sesame harvesting, their inherent trade-offs, and target parameters.
2.3.6 Molecular and genetic regulation of seed shattering resistance in sesame
Advances in genomics are rapidly uncovering the complex regulatory networks that govern shattering resistance. Beyond the specific QTLs and candidate genes mentioned previously, transcriptome-wide studies are revealing broader patterns. These include the differential expression of genes involved in lignin deposition, pectin modification, and cellulose biosynthesis, which collectively reinforce capsule structure (Wang and Brutnell, 2010; Maity et al., 2021). Furthermore, key hormonal pathways–particularly those involving auxin and ethylene–show distinct expression profiles in resistant versus shattering-prone genotypes, indicating their role in regulating abscission zone development (Liu et al., 2024; Wu et al., 2023). Insights from model species like Arabidopsis, rice, and soybean continue to be invaluable for identifying orthologous genes in sesame, accelerating functional validation studies (Wang et al., 2022). As illustrated in Figure 5, a systems-level understanding of these integrated networks is now forming the basis for genomics-assisted breeding of mechanisation-ready sesame.
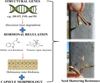 |
Fig. 5 Genetic and hormonal regulation of seed shattering resistance in sesame. The schematic illustrates key molecular and morphological determinants of shattering resistance, including: Structural gene networks (e.g., SHATTERING1 [SHAT1], INDEHISCENT [IND], and Polygalacturonase [PG] genes) controlling cell wall degradation at the abscission layer; hormonal regulation by auxin (inhibiting abscission) and gibberellin (promoting lignin deposition) and capsule morphology traits, such as dehiscence zone thickness and vascular bundle orientation, that mechanically constrain capsule opening. |
3 Mechanised harvesting of sesame
3.1 Mechanised harvesting methods
The transition from traditional manual harvesting to mechanization represents a pivotal step for enhancing sesame production efficiency, but involves navigating significant trade-offs. Manual harvesting, while adaptable to shattering-prone varieties and requiring minimal capital investment, suffers from high labor costs, significant harvest and post-harvest losses (often 15–50% as detailed in Tab. 1), and limitations in scalability. Mechanized harvesting, conversely, offers the potential for drastically reduced labor requirements, faster operations, and lower operational losses if implemented correctly, but faces challenges related to high initial investment, the need for shatter-resistant cultivars with suitable architecture, and precise machine calibration. Therefore, the core strategy to minimize overall losses and maximize economic benefits hinges on successfully bridging the gap between sesame biology and engineering-developing suitable ideotypes (Sect. 2.3.5) and optimizing harvesting technologies (Sect. 4)–which forms the central theme of this review.
Technological progress for sesame mechanization has involved both adapting existing combine harvesters (primarily through header and threshing/cleaning adjustments detailed later) and, to a lesser extent, developing specialized or smaller-scale harvesting equipment tailored to the crop’s specific challenges (Bandhiya et al., 2023, Pari et al., 2020). The mechanised harvesting of sesame is primarily done using two main approaches: (1) two-stage (indirect) harvesting, which involves cutting the plants and allowing them to dry in the field before threshing, and (2) direct harvesting with a combine harvester (Bazyar et al., 2019; Razaghi, 2024a).
In humid or cool regions such as northern Iran (Golestan, Mazandaran), where sesame is sown from late May to mid-June (Gholamhoseini, 2020), ripening often coincides with autumn rains and high humidity, delaying drying and making direct combining impractical due to elevated grain moisture and mechanical risks (Usman et al., 2022). Under such conditions, two-stage harvesting is preferred: plants are cut and windrowed to accelerate drying via improved air circulation and solar exposure (Fig. 6). This lowers seed moisture to safe levels for threshing and storage, and shortens the interval before the next crop, boosting productivity and resource efficiency. However, it requires extra labour and machinery for cutting and threshing, increasing costs compared with direct harvesting under favourable climates (Razaghi, 2024b).
 |
Fig. 6 Natural moisture variation trends in standing sesame plants (for direct harvest method, left image) versus two-stage harvested plants (right image) for whole plants, capsules, and seeds in Golestan Province, Iran. Unpublished data from the authors. |
3.2 Optimal harvest timing: a key factor in minimising losses
Accurate harvest timing is essential to balance seed maturity, minimise shattering, and ensure compatibility with mechanised operations (Neme et al., 2020). Harvesting too early yields immature seeds from apical capsules, reducing yield and quality (Saboury et al., 2021), while delays increase shattering risk and seed moisture, especially under poor weather (Gholamhoseini et al., 2023a).
The optimal window depends on the harvest method (Langham et al., 2010). In two-stage harvesting, plants are cut when ∼ 70−80% of mid-stem capsule seeds are mature-indicated by colour change and a dark hilum–while lower stems yellow and upper stems remain greenish-yellow, allowing remaining seeds to ripen during field drying (Doko and Enwere, 2014). This minimises losses and shortens post-harvest drying. For direct combining, seed moisture should be ∼ 6% for proper threshing (Langham et al., 2010; Benaseer et al., 2018). Field cues include brittle capsules, breakable stems, dust clouds during harvest, and uniform grain flow. Given the potential for moisture variability across a field, ensuring optimal harvest timing may require either staggered harvesting or diligent moisture monitoring. The latter is practically achieved using portable digital moisture meters to confirm that seed moisture has reached the target level of approximately 6% before combining begins (Gholamhoseini, 2023).
4 Direct (one-stage) harvesting of sesame
Direct harvesting (one-stage or combine harvesting) integrates cutting, threshing, and seed collection in a single pass using a combine harvester (Rithiga et al., 2024). Success depends on synchronising plant maturity with optimal field drying (Price et al., 1996). Cultivar traits-particularly capsule shattering resistance and plant architecture–also influence suitability for direct combining (Dossa et al., 2017). The following sections address technical, agronomic, and equipment considerations for one-stage harvesting.
4.1 Complementary plant traits for direct (single-pass) harvest of sesame
In addition to capsule-related traits that constitute the necessary conditions for successful mechanised sesame harvesting, a set of sufficient complementary plant traits can further enhance the efficiency and quality of direct (single-pass) harvest operations. These traits influence key aspects such as seed maturity synchronisation, plant architecture compatibility with combine machinery, and post-harvest seed quality. For instance, uniform maturity is essential for minimising losses from asynchronous ripening, while physical traits must be compatible with the combine’s header. Critical examples include an optimal plant height (120−150 cm) for smooth feeding and a sufficient height to the first capsule (15−30 cm) to allow for a clean cut. These and other key complementary traits are summarised in Table 7.
Complementary morphological and physiological traits contributing to efficient direct sesame harvest.
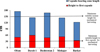 |
Fig. 7 Height to first capsule from ground level, capsule-bearing zone length, and total plant height in Iranian sesame cultivars. Data from Siahpoosh et al. (2024) and Karimi et al. (2019). |
4.2 Optimising combine-harvester dynamics for sesame’s unique biological traits
While most modern combine harvesters can be adapted for sesame, achieving an efficient and high-quality harvest requires a fundamental shift away from standard cereal settings towards a highly specialized calibration. The critical importance of precise calibration is vividly illustrated in Figure 8, which shows the stark difference in the quality of the final product between an optimised and a sub-optimally configured harvester.
The optimisation process begins well before the harvest itself, with a crucial preparatory phase 30-40 days prior, involving thorough cleaning of the harvester with compressed air to eliminate residual grains and weed seeds, and sealing any potential leakage points. This pre-harvest diligence alone can prevent cross-contamination and reduce subsequent processing costs by up to 30% (Razaghi, 2024a and b).
The initial interaction with the crop at the combine’s header is a critical stage where significant losses can occur. To minimise these losses, which can range from 18−22% with improper settings (Sadeghi et al., 2022), the reel speed must be precisely synchronised to be approximately 25% faster than the ground speed (a reel index of 1.0-1.5), making contact with only the top 15 cm of the plant (Elebaid et al., 2016). This gentle handling prevents the violent agitation that causes premature shattering in susceptible genotypes.
The core of the operation–the threshing unit–demands the most significant adjustments. Sesame’s delicate, high-oil seeds are highly susceptible to mechanical damage. Consequently, a much less aggressive threshing action is required compared to hardier grains. This is achieved by using slightly worn serrated cylinders and, most importantly, by reducing the cylinder speed to a low range of 350−500 rpm while widening the concave clearance to 25−30 mm at the front and 12−17 mm at the rear (Ishpekov et al., 2014; Eindhuja et al., 2022). These settings are designed to ensure that seed damage remains below a 2% threshold (Narayanan et al., 2024). An improper balance here can lead to either excessive seed breakage or a high percentage of unthreshed capsules exiting the machine, necessitating a methodical, single-variable adjustment approach for troubleshooting (Kailashkumar, 2021; Razaghi, 2024a). This engineering adjustment is directly dictated by the biological traits described in Section 2.1; specifically, genotypes with high membrane coverage and seed-placenta adhesion scores (providing structural resistance) require a careful increase in cylinder impact force to ensure release, whereas varieties with lower adhesion scores must be harvested at minimum cylinder speeds to prevent seed micro-damage. This necessity to fine-tune machine parameters based on specific capsule histology underscores the critical interdependence of biology and engineering in sesame mechanization.
Finally, the separation and cleaning process must be adapted for sesame’s small seed size (Bandhiya et al., 2023). This involves using fine-mesh sieves (e.g., an upper sieve of 4−12 mm and a lower one of 2−6 mm) and carefully calibrating the fan speed (initially 650−750 rpm) to create an airflow sufficient to remove chaff without blowing the lightweight seeds out of the combine–a common source of significant yield loss (Langham et al., 2010). Even the final stages of grain handling, such as auger and elevator operation, must be managed to avoid damage from sharp edges or excessive initial torque during unloading. The operational efficiency is further modulated by the combine’s forward speed, which must be calibrated based on field size and crop density, ranging from as low as 1.5 km/h in dense stands on small farms (Noorani et al., 2023) to over 11 km/h in sparse stands on large, level fields (Langham et al., 2010). Figure 9 provides a schematic overview summarizing these critical interactions between key capsule traits and the necessary combine harvester parameter adjustments. A summary of the specific recommended settings for these adjustments is provided in Table 8.
 |
Fig. 8 Comparison of harvested sesame yield using a combine harvester with optimised settings (right) versus suboptimal configurations (left). Photo by the authors. |
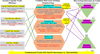 |
Fig. 9 Schematic flow diagram illustrating the critical interactions between sesame capsule traits (biology) and combine harvester settings (engineering) for optimized mechanized harvesting. The diagram highlights how specific capsule characteristics necessitate adjustments in threshing, cleaning, and header units to achieve the desired outcomes (minimized loss, minimized damage, maximized purity), while emphasizing the inherent trade-offs involved in balancing shatter resistance with threshability. |
Key combine harvester adjustments for direct sesame harvesting.
5 Evaluation of sesame yield losses in direct combine harvesting
While the Sesaco and Langham methods are valuable for quantifying a genotype’s intrinsic shatter resistance potential under controlled conditions, they can not replicate the complex and dynamic forces of a real-world harvest. These laboratory-based assays do not account for the cumulative effects of machine-crop interactions-such as reel impact, cutter bar vibrations, and threshing forces-or the influence of variable environmental conditions.
Therefore, field evaluation of seed losses during mechanised harvesting remains essential to assess a genotype’s true performance within a complete agricultural system. Actual machine-induced seed loss during harvesting differs significantly from pre-harvest natural shattering (Loss type 1, Fig. 10), which can exceed 50%insusceptiblevarieties left to dry fully before direct combining (Gholamhoseini and Dolatabadian, 2024a). Focusing on losses during the operation itself, machine related losses are often reported between 5–15% for direct combining (Naydenov et al., 2020; Razaghi, 2024a), where as losses from mechanical handling in two-stage harvesting typically range from 3–10% (Sadeghi et al., 2022; Razaghi, 2024b). During direct combining, these operational losses primarily occur at three key machine points, as illustrated in Figure 10:
Cutting unit losses (Loss type 2): Seed or branch loss caused by cutting blade vibrations, wheel and reel impacts, and partial throwing of branches outside the header.
Unthreshed capsules in combine discharge (Loss type 3): Due to ineffective threshing and separation, whole capsules may exit the combine with the straw.
Losses from the cleaning unit (Loss type 4): Seeds lost through cleaning due to improper settings of the cleaning system and fan airflow.
Therefore, a thorough field assessment must quantify these different loss components, alongside the efficiency of seed release (threshability) and any resulting seed damage. Reliable evaluation of these factors requires plots ≥0.5 ha to ensure stable combine operation and avoid confounding edge effects, and a seed moisture of ∼ 6%, as higher levels impede threshing and increase seed damage (Langham, 2014; Razaghi, 2024a)
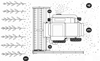 |
Fig. 10 Schematic illustration of seed shattering assessment points during mechanised sesame harvesting. (1) Natural shattering site, (2) Header unit shattering site, (3 & 4) Threshing and cleaning unit shattering sites. Source: Authors. |
5.1 Determination of actual seed yield
Accurate estimation of the actual seed yield (kg ha−1) is essential for assessing losses from natural shattering and mechanised harvesting. To account for spatial variability, sample at least four representative points per hectare (Gholamhoseini, 2022b). At each point, harvest sesame plants from a 1 m2 area using a standard frame and garden shears to avoid disturbance losses (Ishpekov and Stamatov, 2015). Separate capsules on a clean surface, extract seeds manually or with a small thresher and weigh them (in grams). The actual seed yield per hectare is then calculated using the following formula:
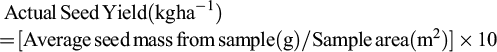
5.2 Estimation of natural shattering losses in the field
Natural seed shattering is the spontaneous seed detachment before mechanical harvesting, causing pre-harvest yield loss (Gan et al., 2008). In sesame, losses depend on genotype-specific capsule traits and environmental stressors such as wind, rain, and humidity fluctuations (Dash et al., 2018). Quantification uses sampling frames placed gently at random field sites (excluding borders) to avoid disturbance. All visible seeds and capsules on the soil surface are collected; if incomplete, losses are estimated from capsule counts and thousand-seed weight (Gan et al., 2016).
5.3 Estimation of cutting unit losses and calculation of harvest loss percentage
This loss category includes seeds dislodged by the combine header during cutting. These losses are particularly high in genotypes sensitive to the mechanical shocks from the reel and the vibrations from the cutter bar (Domeika et al., 2008). For example, traditional Iranian cultivars such as Darab 1 and Dashtestan 2, which are documented as highly shattering in Tables 2 and 4, are prime examples of genotypes susceptible to such header losses. Assessment uses 0.5 m2 seed loss frames (10 cm height, 2 cm wall, fine mesh bottom) placed at four sampling sites–two per site–behind the header at its central and lateral thirds. Frames are positioned to exclude combine discharge and external seeds, with any plant parts extending inward trimmed (Eckert et al., 2011). After harvest, free seeds and seeds from manually threshed capsules within frames are weighed. In addition, to express measured seed losses as percentages of yield, the following formula is used:
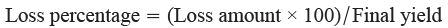
5.4 Assessment of seed release efficiency from capsules
Assessing seed release efficiency, a direct measure of a genotype’s threshability, is critical as it impacts both yield and quality. Inefficient release leads to direct yield loss through unthreshed capsules and can indirectly degrade seed quality, as operators may resort to more aggressive, damaging combine settings to compensate. To assess seed release efficiency during mechanised harvesting, a comparative capsule-counting method is used (Razaghi, 2024a and b):
Mechanically harvested capsules – Randomly collect 100 partially or unthreshed capsules from the combine’s output (often from frames used in threshing loss assessment) and count remaining seeds.
Reference capsules – Randomly select 20 plants from the field interior (≥50−100 m from edges); pick five mature capsules per plant from various positions (excluding immature apical capsules) and count total seeds from these 100 capsules. The seed release percentage is calculated as follows:

5.5 Evaluation of seed damage during mechanised harvesting
Seed damage is a key quality metric in mechanised sesame harvesting (Narayanan et al., 2025). To assess it, harvesting continues until the grain tank is nearly full. Samples are taken from four positions and depths within the tank, combined, and mixed into a 60 g composite (Wang et al., 2021). Seeds are classified as: (1) intact, (2) damaged/broken, and (3) immature seeds, weed seeds, and foreign material. For damage estimation, only the first two categories are used. After separating and weighing intact and broken seeds, the damage percentage is calculated as:
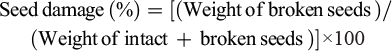
A summary of the key procedures and measurements described in Section 5 is presented in Table 9.
Summary of key parameters and methods for evaluating seed loss and post-harvest quality in mechanised sesame harvesting.
5.6 Standards for seed shattering resistance in mechanised sesame harvesting
According to the framework proposed by Langham (2014) and Gholamhoseini (2022b), a sesame genotype can be considered resistant to shattering and suitable for direct combine harvesting if it meets the following four criteria:
At least 85% of seeds must remain within the capsules until the harvest date, when seed moisture content has declined to 6% or less (i.e., natural seed shattering <15%).
Maximum 7% seed loss should occur during direct combine harvesting (header and cleaning units combined).
Maximum 5% of seeds may remain unreleased in the capsules after harvest.
Seed damage due to mechanised harvest must not exceed 3%.
These quantitative thresholds serve as an operational benchmark for evaluating breeding lines and assessing the field readiness of mechanised sesame harvest systems.
6 Quality control of mechanically harvested sesame seed
Maintaining sesame seed quality after mechanical harvest is essential for domestic and export markets (Angamuthu et al., 2025). Quality standards should be followed throughout production, harvest, and post-harvest processes (Neme et al., 2020). While no official national sesame standards exist in Iran or many major producing countries, internationally recognised benchmarks define key export parameters (Tab. 10).
Untimely harvest, particularly at high seed moisture, reduces quality (Gholamhoseini, 2023). Field-wide moisture assessment and avoiding high-humidity hours (early morning/late evening) are crucial. Full physiological maturity is required, as late planting or cold during seed filling increases immature seeds that must be removed post-harvest (Gholamhoseini and Dolatabadian, 2024a). Foreign materials–broken seeds, weed seeds, soil, stones, debris, or insects—lower market value and raise moisture and spoilage risk (Peariso, 2008). Weed seeds are a particularly problematic foreign material as they also spread infestations (Duary, 2014); control measures range from in-field management and pre-harvest combine cleaning to segregating weedy areas during harvest and utilizing advanced cleaning systems (e.g., optical sorters) post-harvest (Gholamhosseini and Mansouri, 2024). Proper adjustment of airflow, sieves, and fans minimises these contaminants overall.
Seed damage mainly stems from incorrect combine settings. While optimally configured systems aim for damage rates below 3.5%, improper settings can easily cause mechanical damage exceeding 10%, significantly reducing market value (Razaghi, 2024a). Causes include excessive threshing speed, narrow concave clearance, overfilled grain tanks, and harvesting above 6% moisture (Razaghi, 2024b). Sharp edges on threshing or conveying parts can also harm oil-rich seeds. Damaged and moist seeds deteriorate quickly, making correct combine adjustment vital for quality assurance (Afzal et al., 2019). Achieving high-quality mechanically harvested sesame therefore requires an integrated approach, managing factors from field conditions (maturity, weeds, moisture) through to precise combine calibration (settings, speed) and careful post-harvest handling. Adherence to internationally recognized benchmarks, such as those summarized in Table 10, is crucial for market access and profitability.
Internationally recognised quality standards for graded sesame seed lots based on key physical and chemical attributes.
7 Postharvest storage and preservation of sesame seeds
Characterized by a high oil content that can exceed 50% (Balouchi et al., 2023), sesame seeds are consequently highly susceptible to quality loss during storage. Proper postharvest handling and storage are essential to preserve quality and protect the crop’s economic value, given its wide uses from direct consumption and oil extraction to value-added products (Osei-Kwarteng and Ogwu, 2024).
7.1 Optimal storage conditions
Sesame seed quality depends heavily on storage temperature and relative humidity (RH) (Adebisi et al., 2008). Lower temperatures prolong shelf life and reduce microbial and enzymatic activity (Gholamhosseini and Mansouri, 2024). Optimal storage is 4−10° C, with further reduction (e.g., 5°C at 40% RH) needed when RH exceeds 25% to prevent fungal growth and biochemical degradation (Sadeghi Germaroodi et al., 2022). Continuous monitoring of temperature and humidity, plus adequate ventilation in bulk storage, is vital. Drying seeds to ≤6% moisture before storage is a critical step for preserving oil quality, as it lowers the seed’s water activity to inhibit fungal growth and inhibits lipase enzyme activity, thereby preventing hydrolytic rancidity (Abdiani et al., 2024)
7.2 Prevention of pest and fungal infestation
Post-harvest losses from insect infestation in sesame can be substantial, with key pests such as the red flour beetle (Tribolium castaneum) and the cigarette beetle (Lasioderma serricorne) causing direct weight losses that can range from 5% to over 20% under suboptimal storage conditions (Berhe et al., 2023, 2024). Furthermore, fungal contamination, particularly by mycotoxin-producing species like Aspergillus flavus, poses a significant health risk (Khan, 2024). To mitigate these threats, fumigation with phosphine (generated from aluminium phosphide) is a widely adopted chemical control measure, which has demonstrated high efficacy, often exceeding 95–98% mortality against susceptible populations of major storage pests when applied at standard dosages (e.g., 1−3 g/m3) for an adequate exposure period (Chaudhry, 1997; Nayak et al., 2020). As a non-chemical alternative, gamma irradiation has also proven effective (Gholamhosseini and Mansouri, 2024). Research indicates that dosages between 0.5 and 1.0 kGy are sufficient for complete disinfestation of stored sesame seeds, while doses up to 5 kGy can control fungal growth without significantly compromising seed viability or oil quality (Hassan et al., 2018; Al-Bachir, 2016).
7.3 Structural considerations
Structural storage practices are critical for maintaining seed quality by managing micro-environmental factors like moisture and pressure. Stacking bags directly on concrete floors, for instance, can lead to moisture wicking, potentially increasing the seed moisture content in the bottom layers by 2−3 percentage points above the safe threshold, creating localized hotspots for fungal proliferation (Gebregergis et al., 2024). Therefore, stacking on pallets with at least a 1 m clearance from walls is essential for preventing moisture transfer and promoting air circulation (Alemayehu et al., 2023). Furthermore, excessive stack height exerts significant compression force on the lower bags. Studies on oilseeds have demonstrated that stack heights exceeding 3 meters can cause mechanical damage, leading to a reduction in germination capacity by as much as 10–15% in the bottom layers and an increase in free fatty acid content due to cracked seed coats (Abdiani et al., 2024). Proper labelling and segregation of seed lots by harvest date and quality grade remain crucial for effective inventory management and preventing cross-contamination.
7.4 Impact on oil quality
Two key indicators of oil degradation during storage are free fatty acid (FFA) content and peroxide value (PV) (Anwar et al., 2007). Increases in these indices indicate hydrolytic and oxidative spoilage, respectively, reducing crude oil quality, refining efficiency, and the flavour, shelf life, and safety of the final product (Tan et al., 2017). Maintaining low seed moisture, avoiding mechanical damage, and preventing microbial growth are essential to preserve oil quality. Furthermore, the initial integrity of the capsule itself, a factor influenced by shatter resistance traits, can play a role by providing a natural physical barrier that protects seeds from environmental fluctuations and pest ingress prior to threshing, potentially contributing to better initial oil quality entering storage.
7.5 Special considerations
Dehulled sesame seeds are more prone to oxidative rancidity due to their increased surface area (Abou-Gharbia et al., 1997); therefore, unhulled seeds are preferred for long-term storage. Processing delays should be minimised, and storage ideally conducted in hermetically sealed containers under controlled atmospheres when possible.
8 Challenges, innovations, and future directions in sesame harvesting and postharvest handling
Sesame production faces growing pressure for mechanisation and reduced postharvest losses, particularly from seed shattering (Myint et al., 2020; Gholamhoseini, 2023; Gholamhoseini and Dolatabadian, 2024a). The biological complexity of capsule dehiscence and plant fragility make direct mechanical harvesting challenging. Progress in breeding shatter-resistant genotypes and refining harvest equipment has been notable but remains underutilised, especially in smallholder systems.
A major constraint is the mismatch between current cultivars and conventional combine harvester requirements, leading to high seed loss and reduced quality. Bridging this gap demands breeding ideotypes and engineering harvesters with sesame-specific adaptive components, requiring collaboration among agronomists, breeders, and engineers.
Future mechanisation will hinge on innovations that are both advanced and socioeconomically accessible. Promising developments–marker-assisted selection, AI-based harvest timing, optical seed sorters, and self-calibrating combines–remain out of reach for many low-input producers. Technologies must be efficient, affordable, adaptable to diverse agroecologies, and supported by local capacity building.
Postharvest quality management is equally critical. Without proper drying, handling, and storage, mechanised harvesting can cause contamination, discolouration, and oil quality loss. Standardised grading, operator training, and alignment with international quality benchmarks are essential for transforming sesame into a high-value crop.
Research priorities include high-throughput phenotyping for shattering traits, precision agriculture tools (drones, ground sensors) for harvest timing, and low-cost modular harvesters for smallholder fields in Africa, South Asia, and the Middle East. Novel strategies, such as bio-based capsule-strengthening agents or temporary surface coatings to reduce mechanical stress, also warrant exploration.
9 Conclusion
Sesame stands at a pivotal juncture, rich in nutritional value, ecological versatility, and economic promise, yet its full potential remains largely untapped. Addressing the technical challenges of seed shattering and mechanised harvesting is essential, but it is not sufficient. As this review has emphasised, sustainable expansion of sesame cultivation demands a systemic transformation that integrates genetic improvement, engineering solutions, and postharvest technologies within a broader socio-economic framework. Mechanisation must be coupled with robust seed systems, farmer-centred knowledge dissemination, infrastructure investment, and supportive institutional environments. Unlocking its future as a resilient, high-value crop requires nothing less than a holistic, inclusive, and strategic realignment of the entire production and value chain. Specifically for smallholder farmers, adopting such mechanisation-ready ideotypes could drastically reduce dependency on costly and often scarce manual labour, improve harvest timeliness, minimise post-harvest losses, and ultimately enhance profitability and resilience, facilitating their better integration into modern value chains. Realizing this potential will require targeted next steps, including establishing pilot breeding programs focused on these ideotypes, conducting participatory on-farm trials to validate performance across diverse environments, and fostering public-private partnerships to facilitate the co-development and dissemination of adapted machinery and cultivars.
Conflicts of interest
The authors declare no conflicts of interest related to this work. All authors confirm that there are no personal, professional, or financial relationships that could potentially influence the results or interpretation of this study. All co-authors have reviewed and approved this statement.
References
- Abebe TN. 2016. Review of sesame value chain in Ethiopia. Int J Afr Asian Stud 19: 36–47. [Google Scholar]
- Abdiani N, Kolahi M, Javaheriyan M, Sabaeian M. 2024. Effect of storage conditions on nutritional value, oil content, and oil composition of sesame seeds. J Agric Food Res 16: 101117. [Google Scholar]
- Abou-Gharbia HA, Shahidi F, Adel A, Shehata Y, Youssef MM. 1997. Effects of processing on oxidative stability of sesame oil extracted from intact and dehulled seeds. J Am Oil Chem Soc 74: 215–221. [Google Scholar]
- Adebisi MA, Ola JA, Akintobi DAC, Daniel IO. 2008. Storage life of sesame (Sesamum indicum L.) seeds under humid tropical conditions. Seed Sci Technol 36(2): 379–387. [Google Scholar]
- Afzal I, Zahid S, Mubeen S. 2019. Tools and techniques of postharvest processing of food grains and seeds. Agronomic Crops: Volume 2: Management Practices, pp. 583–604. [Google Scholar]
- AGMARK Standards. 2025. Grade designation and definition of quality of sesame seeds. Retrieved from https://paperzz.com/doc/7826056/sesame---agri-exchange [Google Scholar]
- Al-Bachir M. 2016. Some microbial, chemical and sensorial properties of gamma irradiated sesame (Sesamum indicum L.) seeds. Food Chem 197: 191–197. [Google Scholar]
- Alemayehu S, Abera FA, Ayimut KM, et al. 2023. Effects of storage duration and structures on sesame seed germination, mold growth, and mycotoxin accumulation. Toxins 15(1): 39. [Google Scholar]
- Andargie M, Vinas M, Rathgeb A, Möller E, Karlovsky P. 2021. Lignans of sesame (Sesamum indicum L.): a comprehensive review. Molecules 26(4): 883. [Google Scholar]
- Angamuthu M, Kasirajan S, Pandurangan G, Langyan S, Perumal V. Organic production of sesame: status, techniques, and market. In Sesame: sustainable production and applications. Singapore: Springer Nature Singapore, 2025, pp. 81–105. [Google Scholar]
- Anwar F, Chatha SAS, Hussain AI. 2007. Assessment of oxidative deterioration of soybean oil at ambient and sunlight storage. Grasas y aceites, 58(4): 390–395. [Google Scholar]
- Ashri A. 1998. Sesame breeding. Plant Breed Rev 16: 179–228. [Google Scholar]
- Ashri A. Sesame (Sesamum indicum L.). In: Singh RJ, ed. Genetic resources, chromosome engineering, and crop improvement, vol. 4, 2006, pp. 231–289. [Google Scholar]
- Ataei R, Gholamhoseini M, Mohammadi V. 2017. Association mapping in plants. Crop Biotechnol 7(1): 25–37. [Google Scholar]
- Balouchi H, Soltani Khankahdani V, Moradi A, et al. 2023. Seed fatty acid changes germination response to temperature and water potentials in six sesame (Sesamum indicum L.) cultivars: estimating the cardinal temperatures. Agriculture 13(10): 1936. https://doi.org/10.3390/agriculture13101936 [Google Scholar]
- Bandhiya R, Gaadhe S, Chavda S, Gojiya K, Gojiya DK, Chavda D. 2023. Cultivation of sesame crop in mechanized way. Int Res J Modern Eng Tech Sci 5: 1455–1461. [Google Scholar]
- Bazyar P, Jafari A, Alimardani R, Mohammadi V. 2019. Small-scale head of combine for harvesting sesame. Agric Eng 22(4): 02. [Google Scholar]
- Bedigian D. 2004. History and lore of sesame in Southwest Asia. Econ Botany 58(3): 329–353. [Google Scholar]
- Bedigian D. ed. 2010. Sesame: the genus Sesamum. CRC press. [Google Scholar]
- Bell B. 2010. Farm machinery. Old Pond Books. [Google Scholar]
- Beech DF, Imrie BC. 2001. Breeding for mechanized sesame production in Australia. In: Van Zanten L, ed. Sesame improvements by induced mutations. Proceedings of the Final FAO/IAEA Co-ordinated Research Meeting, IAEA. (TECDOC-1195), pp. 63–70 [Google Scholar]
- Benaseer S, Masilamani P, Albert VA, Govindaraj M, Selvaraju P, Bhaskaran M. 2018. Impact of harvesting and threshing methods on seed quality-a review. Agric Rev 39(3): 183–192. [Google Scholar]
- Berhe M, Subramanyam B, Demissie G, et al. 2023. Effect of storage duration and storage technologies on pest infestations and post-harvest quality loss of stored sesame seeds in Ethiopia. J Stored Prod Res 103: 102161. [CrossRef] [Google Scholar]
- Berhe M, Subramanyam B, Demissie G, et al. 2024. Insect species dynamics and associated losses in on-farm stored sesame (Sesamum indicum L.) seeds in major sesame growing areas in Ethiopia. Int J Trop Insect Sci 44(2): 855–871. [Google Scholar]
- CBI Market Information. 2025. Entering the European market for sesame seeds. https://www.cbi.eu/market-information/grains-pulses-oilseeds/sesame-seeds/market-entry [Google Scholar]
- Chaudhry MQ. 1997. Review a review of the mechanisms involved in the action of phosphine as an insecticide and phosphine resistance in stored-product insects. Pestic Sci 49(3): 213–228. [Google Scholar]
- Chinese Standard. 2025. Sesame seed – National Standard of the People’s Republic of China. https://www.chinesestandard.net/PDF/English.aspx/GBT11761-2021?Redirect [Google Scholar]
- Dash M, Imran M, Kabi M, Baisakh B, Lenka D. 2018. Assessment of genetic variability for capsule shattering characters in Indian sesame. Electron J Plant Breed 9(2): 490–501. [Google Scholar]
- Day JS. 2000a. Development and maturation of sesame seeds and capsules. Field Crops Res 67: 1–9. [Google Scholar]
- Day JS. 2000b. Anatomy of capsule dehiscence in sesame varieties. J Agric Sci 134(1): 45–53. [Google Scholar]
- Day JS. 2000c. The effect of plant growth regulator treatments on plant productivity and capsule dehiscence in sesame. Field Crops Res 66(1): 15–24. [Google Scholar]
- Doko B, Enwere S. 2014. Farmer’s guide for the production and post-harvest handling of sesame products in Nigeria. Training Manual 54. [Google Scholar]
- Domeika R, Jasinskas A, Steponavičius D, Vaiciukevičius E, Butkus V. 2008. The estimation methods of oilseed rape harvesting losses. Agron Res 6: 191–198. [Google Scholar]
- Dong Y, Wang YZ. 2015. Seed shattering: from models to crops. Front Plant Sci 6: 476. [Google Scholar]
- Dong Y, Yang X, Liu J, Wang BH, Liu BL, Wang YZ. 2014. Pod shattering resistance associated with domestication is mediated by a NAC gene in soybean. Nat Commun 5(1): 3352. [Google Scholar]
- Dossa K, Diouf D, Wang L, et al. 2017a. The emerging oilseed crop Sesamum indicum enters the “Omics” era. Front Plant Sci 8: 1154. [Google Scholar]
- Dossa K, Konteye M, Niang M, Doumbia Y, Cissé N. 2017b. Enhancing sesame production in West Africa’s Sahel: A comprehensive insight into the cultivation of this untapped crop in Senegal and Mali. Agric Food Sec 6: 1–15. [Google Scholar]
- Dossa K, Li D, Wang L, et al. 2018. Genome-wide association studies of 39 seed yield-related traits in sesame (Sesamum indicum L.). Int J Mol Sci 19(9): 2794. [Google Scholar]
- Dossa KF, Enete AA, Miassi YE, Omotayo AO. 2023. Economic analysis of sesame (Sesamum indicum L.) production in Northern Benin. Front Sustain Food Syst 6: 1015122. [Google Scholar]
- Duary B. 2014. Weed prevention for quality seed production of crops. SATSA Mukhapatra-Annu Tech (18): 48–57. [Google Scholar]
- Dutta D, Banerjee S, Pal M. 2022. Validation of determinate (dt) gene-based DNA marker in inter-specific hybrid sesame and in-silico analysis of the predicted dt protein structures. Physiol Mol Biol Plants 28: 139–152. [Google Scholar]
- Eckert FR, Kandel HJ, Johnson BL, et al. 2011. Seed yield and loss of dry bean cultivars under conventional and direct harvest. Agron J 103(1): 129–136. [Google Scholar]
- Eindhuja M, Giridharan K, Kamali K, Sakthi Vignesh S. 2022. Design and development of sesame threshing unit. Sci Hub Appl Res Eng Inf Technol 2(3): 19–26. [Google Scholar]
- Elebaid JI, Abdalla SA, Osama AM. 2016. Performance evaluation of self-propelled sesame harvesting cutter binder under Gedarif farming conditions, Gedarif State, Sudan. RUFORUM working document series (ISSN 1607-9345), 14(2): 1085–1094. [Google Scholar]
- Food and Agriculture Organization of the United Nations. 2025. FAOSTAT Database: Production – Crops and livestock products [Data set]. Retrieved October 25, 2025, Available at: https://www.fao.org/faostat/en/#data/QCL [Google Scholar]
- Fuller DQ, Allaby R. 2009. Seed dispersal and crop domestication: shattering, germination and seasonality in evolution under cultivation. Annual plant reviews volume 38: fruit development and seed dispersal, 38: 238–295. [Google Scholar]
- Gan Y, Malhi SS, Brandt SA, McDonald CL. 2008. Assessment of seed shattering resistance and yield loss in five oilseed crops. Can J Plant Sci 88(1): 267–270. [Google Scholar]
- Gan Y, Blackshaw RE, May WE, Vera C, Johnson EN. 2016. Yield stability and seed shattering characteristics of Brassica juncea canola in the Northern Great Plains. Crop Sci 56(3): 1296–1305. [Google Scholar]
- Gebregergis Z, Baraki F, Fiseseha D. 2024. Effects of environmental factors and storage periods on sesame seed quality and longevity. CABI Agric Biosci 5(1): 1–11. [Google Scholar]
- Gebremichael DE. 2017. Sesame (Sesamum indicum L.) breeding in Ethiopia. Int J Novel Res Life Sci 4(1): 1–1. [Google Scholar]
- Gebretsadik D, Haji J, Tegegne B. 2019. Sesame post-harvest loss from small-scale producers in Kafta Humera District, Ethiopia. J Dev Agric Econ 11(2): 33–42. [Google Scholar]
- Gholamhoseini M. 2020. Evaluation of sesame genotypes for agronomic traits and stress indices grown under different irrigation treatments. Agron J 112: 1794–1804. [CrossRef] [Google Scholar]
- Gholamhoseini M. 2022a. Optimizing irrigation and nitrogen fertilization of Iranian sesame cultivars for grain yield and oil quality. J Food Compos Anal 108: 104448. [Google Scholar]
- Gholamhoseini M. 2022b. Determining the optimal planting density and arrangement for a shatteringtolerant sesame genotype in the major producing provinces (Final research report No. 62808, 64 pp.). Seed and Plant Improvement Institute. [In Persian] [Google Scholar]
- Gholamhoseini M, Mansouri S, Masoudi B, Shariati F. 2022a. Evaluation of grain yield and agronomic traits of foreign sesame (Sesamum indicum L.) genotypes under drought stress conditions. J Crop Prod Process 12(2): 19–33. [Google Scholar]
- Gholamhoseini M, Babaei HR, Mansouri S, Shariati F. 2022b. Effect of planting arrangement and plant density on seed yield of shattering-tolerant sesame (Sesamum indicum L.) genotype in Karaj and Mashhad in Iran. Seed Plant J 38(3): 283–303. [Google Scholar]
- Gholamhoseini M. 2023. Current status and roadmap for sesame production in Iran (Technical Bulletin No. 64784, 43 pp.). Seed and Plant Improvement Institute. [In Persian] [Google Scholar]
- Gholamhoseini M, Peyghamzadeh K, Mansouri S, Faraji A, Shariati F. 2023a. Performance evaluation of imported seed-shattering resistant sesame genotype under different planting arrangements and densities in Gorgan. J Crop Prod 16(4): 133–148. [Google Scholar]
- Gholamhoseini M, Ghodrati G, Alhani A, Mansori S, Shariati F. 2023b. Evaluation of yield performance of a shattering-tolerant sesame genotype under different planting arrangements in the Darab and Dezful region. Plant Prod 46(2): 293–305. [Google Scholar]
- Gholamhosseini M, Mansouri S. 2024. Technical guidelines for sesame seed production (Technical Bulletin No. 65244, 36 pp.). Seed and Plant Improvement Institute, Iran. [In Persian] [Google Scholar]
- Gholamhoseini M, Dolatabadian A. 2024a. Sesame renaissance: Iran’s emerging influence in the global sesame market. OCL, 31: 24. [Google Scholar]
- Gholamhoseini M, Dolatabadian A. 2024b. Sesame germination dynamics: Unravelling sesame’s response to salinity and temperature variability. Seeds 3(1): 76–87. [Google Scholar]
- Gholamhoseini M, Zeinalzadeh-Tabrizi H, Abbasali Andarkhor S, Mansouri S, Shariati F, Parchami-Araghi F. 2024a. The effect of planting arrangement and plant density on the yield of non-dehiscent sesame in Sari and Moghan. J Plant Prod Res 31(1): 171–188. [Google Scholar]
- Gholamhoseini M, Aein A, Bakhshi B, Mansouri S, Shariati F. 2024b. Yield evaluation of a shattering-tolerant sesame genotype grown under different planting arrangements and densities in the hot and dry climate of the South East. Crop Sci Res Arid Regions 6(2): 413–430. [Google Scholar]
- Grand View Research. 2024. Sesame Seeds Market Size, Share & Trends Analysis Report By Application (Food, Pharmaceuticals), By Form (Raw/Whole, Processed), By Region, And Segment Forecasts, 2024-2030. Grand View Research, Inc. Available at: https://www.grandviewresearch.com/industry-analysis/sesame-oil-market-report [Google Scholar]
- Habibzadeh F, Gholamhoseini M. 2022. Selection of the best planting method and plant density for two sesame (Sesamum indicum L.) cultivars with different growth types in Karaj region. J Plant Prod Res 29(1): 191–207. [Google Scholar]
- Hamedani NG, Gholamhoseini M, Bazrafshan F, Habibzadeh F, Amiri B. 2022. Yield, irrigation water productivity and nutrient uptake of arbuscular mycorrhiza inoculated sesame under drought stress conditions. Agric Water Manag 266: 107569. [Google Scholar]
- Hassan AB, Mahmoud NS, Elmamoun K, Adiamo OQ, Ahmed IAM. 2018. Effects of gamma irradiation on the protein characteristics and functional properties of sesame (Sesamum indicum L.) seeds. Radiat Phys Chem 144: 85–91. [Google Scholar]
- Ishpekov S, Petrov P, Triffonov A, et al. 2014. Investigation of the indices for mechanized picking sesame capsules. Bulg J Agric Sci 18: 628–633. [Google Scholar]
- Ishpekov S, Stamatov S. 2015. Method for assessment the susceptibility of sesame genotypes for mechanized harvesting of the seed. Bulg J Agric Sci 21(6): 1230–1233. [Google Scholar]
- Ishpekov S, Stamatov S. 2019. Overview of sesame research in Bulgaria. Mech Agric Conserv Resour 65(1): 17–24. [Google Scholar]
- Ju M, Li G, Tian Q, et al. 2024. Deletion of a 1,049 bp sequence from the 5′ UTR upstream of the SiHEC3 gene induces a seed non-shattering mutation in sesame. J Integr Agric 23(8): 2589–2604. [Google Scholar]
- Kailashkumar EB. 2019. A need for sesame thresher. Int J Trend Sci Res Dev 3: 462–464. [Google Scholar]
- Kailashkumar EB. 2021. Development and evaluation of a sesame thresher as influenced by crop, machine and operational parameters. J Appl Nat Sci 13. [Google Scholar]
- Karimi Z, Aghaalikhani M, Gholamhoseini M. 2019. Study of planting density on agronomic traits of sesame cultivars. Iran J Field Crops Res 16(4): 821–831. [Google Scholar]
- Kefale H, Wang L. 2022. Discovering favorable genes, QTLs, and genotypes as a genetic resource for sesame (Sesamum indicum L.) improvement. Front Genet 13: 1002182. [Google Scholar]
- Kelali K, Misikir M, Kormelinck A. 2014. Sesame yields and post-harvest losses in Ethiopia: evidence from the field. Genome Biol 15: 1–13. [CrossRef] [Google Scholar]
- Khan AA. 2024. Insect as major carrier of aflatoxins and mycotoxin in foods: a review. J Entomol Zool Stud 12(3): 46–53. [Google Scholar]
- Langham DR. 2000. Method for making non-dehiscent sesame (United States Patent No. 6, 100, 452). [Google Scholar]
- Langham DR, Wiemers T. 2002. Progress in mechanizing sesame in the US through breeding. In: Janick J, ed. Trends in new crops and new uses. ASHS Press, pp. 157–173. [Google Scholar]
- Langham DR. 2007. Phenology of sesame. In: Janick J, Whipkey A, eds. Issues in new crops and new uses. ASHS Press, pp. 144–182. [Google Scholar]
- Langham DR, Riney J, Smith G, Wiemers T. 2008. Sesame grower guide. Sesaco Corp 30(331): 3. [Google Scholar]
- Langham DR, Riney J, Aubin J, et al. 2010. Sesame harvest guide. Sesaco Corporation. [Google Scholar]
- Langham DR. 2011. Non-dehiscent sesame (United States Patent No. 8,080,707). [Google Scholar]
- Langham DR. 2013. Method for breeding improved non-dehiscent sesame (United States Patent No. 8,581,028 B2). [Google Scholar]
- Langham DR. 2014. Pygmy sesame plants for mechanical harvest (United States Patent No. 8,664,472). [Google Scholar]
- Li LF, Olsen KM. 2016. To have and to hold: selection for seed and fruit retention during crop domestication. Curr Topics Dev Biol 119: 63–109. [Google Scholar]
- Li H, Tahir ul Qamar M, Yang L, Liang J, You J, Wang L. 2023. Current progress, applications and challenges of multi-omics approaches in sesame genetic improvement. Int J Mol Sci 24(4): 3105. [Google Scholar]
- Lima DDC, Dutra AS, Camilo JDM. 2014. Physiological quality of sesame seeds during storage. Revista Ciência Agronômica 45: 138–145. [Google Scholar]
- Liu L, Javed HH, Hu Y, Luo YQ, Peng X, Wu YC. 2024. Research progress and mitigation strategies for pod shattering resistance in rapeseed. PeerJ 12: e18105. [Google Scholar]
- Maity A, Lamichaney A, Joshi DC, et al. 2021. Seed shattering: a trait of evolutionary importance in plants. Front Plant Sci 12: 657773. [Google Scholar]
- Miao H, Langham DR, Zhang H. 2021. Botanical descriptions of sesame. In: The sesame genome. Springer, pp. 19–57. [Google Scholar]
- Myint D, Gilani SA, Kawase M, Watanabe KN. 2020. Sustainable sesame (Sesamum indicum L.) production through improved technology: an overview of production, challenges, and opportunities in Myanmar. Sustainability 12(9): 3515. [Google Scholar]
- Narayanan R, Bhattarai S, Tram VH, Hunter L. 2024. Adapting grain crop seed threshing equipment for processing sesame for streamlining the mechanisation of post-harvest operations. In: IOP Conference Series: Earth and Environmental Science. IOP Publishing, Vol. 1290, No. 1, p. 012015. [Google Scholar]
- Narayanan R, Tram VH, Trotter T, et al. 2025. Adaptation of grain cleaning equipment for kalonji and sesame seeds. AgriEngineering 7(6): 179. [Google Scholar]
- Nayak MK, Daglish GJ, Phillips TW, Ebert PR. 2020. Resistance to the fumigant phosphine and its management in insect pests of stored products: a global perspective. Ann Rev Entomol 65(1): 333–350. [Google Scholar]
- Naydenov N, Ishpekov S, Zaykov R. 2020. Investigation of seed losses at mechanized harvesting of sesame. Bulg J Agric Sci 26: 1323–1327 [Google Scholar]
- Neme K, Tola YB, Mohammed A, Tadesse E. 2020. Postharvest handling practices and on-farm estimation of losses of sesame seeds (Sesamum indicum L.): the case of two Wollega zones in Ethiopia. East Afr J Sci 14(1). [Google Scholar]
- Noorani MH, Asakereh A, Siahpoosh MR. 2023. Evaluation of mechanized and traditional harvesting methods of sesame in the north of Khuzestan province. J Agric Mech 8(1): 33–42. [Google Scholar]
- Nyo HT, Htwe NN, Win KK. 2021. Measurement of postharvest losses of sesame (Sesamum indicum L.) from harvesting to storage at farm level. Int J Environ Rural Dev 12(2): 223–229. [Google Scholar]
- Osei-Kwarteng M, Ogwu MC. 2024. Post-harvest food quality and safety. Food Safety Qual Global South 151. [Google Scholar]
- Pari L, Latterini F, Stefanoni W. 2020. Herbaceous oil crops, a review on mechanical harvesting state of the art. Agriculture 10(8): 309. [Google Scholar]
- Pathak N, Rai AK, Kumari R, Bhat KV. 2014. Value addition in sesame: a perspective on bioactive components for enhancing utility and profitability. Pharmacognosy Rev 8(16): 147. [Google Scholar]
- Peariso D. 2008. Preventing foreign material contamination of foods. John Wiley & Sons. [Google Scholar]
- Price JS, Hobson RN, Neale MA, Bruce DM. 1996. Seed losses in commercial harvesting of oilseed rape. J Agric Eng Res 65(3): 183–191. [Google Scholar]
- Phumichai C, Matthayatthaworn W, Chuenpom N, et al. 2017. Identification of a scar marker linked to a shattering resistance trait in sesame. Turkish J Field Crops 22(2): 258–265. [Google Scholar]
- Queiroga VP, Langham DR, Gomes JP, Melo BA, Santos YMG, Albuquerque EMB. 2019. Technologies used in the cultivation of mechanized sesame (Sesamum indicum L.). Barriguda Publication. [Google Scholar]
- Qureshi M, Langham DR, Lucas SJ, Uzun B, Yol E. 2022. Breeding history for shattering trait in sesame: classic to genomic approach. Mol Biol Rep 49(7): 7185–7194. [Google Scholar]
- Rademacher FJC. 1981. On seed damage in grain augers. J Agric Eng Res 26(1): 87–96. [Google Scholar]
- Rithiga R, Natarajan SK, Venkatachalam SR, Rathika S, Sivakumar R. 2024. Complete mechanization of oil seed crop cultivation to augment the potentiality of yellow revolution in india: a review. Int J Plant Soil Sci 36(8): 550–569. [Google Scholar]
- Razaghi MH. 2024a. Appropriate harvesting method for shattering-resistant sesame to produce high-quality seed (Final Research Report No. 10108, 79 pp.). Agricultural Engineering Research Institute. [In Persian] [Google Scholar]
- Razaghi MH. 2024b. Investigation of desiccants’ effects on seed quantity and quality in mechanized harvest of shattering-tolerant sesame (Final Research Report No. 10107, 93 pp.). Agricultural Engineering Research Institute. [In Persian] [Google Scholar]
- Saboury A, Gholamhoseini M, Bazrafshan F, Habibzadeh F, Amiri B. 2021. Interaction of irrigation and nitrogen fertilization on yield and input use efficiency of sesame cultivars. Agron J 113: 5133–5142. [CrossRef] [Google Scholar]
- Sadeghi GH, Gholamhoseini M, Habibzadeh F. 2022. Sesame production: Challenges and solutions. Imam Khomeini International University Press. [In Persian] [Google Scholar]
- Sesaco. 2025. About Us. Retrieved from https://sesaco.com/about-us-2 [Google Scholar]
- Shahid LA, Saeed MA, Amjad N. 2010. Present status and future prospects of mechanized production of oilseed crops in Pakistan-a review. Pak J Agric Res 23(1-2). [Google Scholar]
- Shtein I, Elbaum R, Bar-On B. 2016a. The hygroscopic opening of sesame fruits is induced by a functionally graded pericarp architecture. Front Plant Sci 7: 1501. [Google Scholar]
- Shtein I, Shelef Y, Marom G, Bar-On B, Elbaum R. 2016b. Gradient of structural traits drives hygroscopic movements of Sesamum indicum capsules. Annals Botany, 119(8): 1365–1372. [Google Scholar]
- Siahpoosh MR, Nejadsadeghi L, Siahpoosh MS, Sheidaie S, Rezvani E, Sadeghi H. 2024. Introduction and evaluation of agronomic features of commercial non-shattering (indehiscent) sesame cultivars named Barkat, Mohajer, Chamran and Dezful. Plant Prod 46(4): 473–490. [Google Scholar]
- Singh TP. 2016. Farm machinery. PHI Learning Pvt. Ltd. [Google Scholar]
- Srivastava AK, Goering CE, Rohrbach RP, Buckmaster DR. 1993. Engineering principles of agricultural machines. American Society of Agricultural Engineers. [Google Scholar]
- Tan CH, Ariffin AA, Ghazali HM, Tan CP, Kuntom A, Choo ACY. 2017. Changes in oxidation indices and minor components of low free fatty acid and freshly extracted crude palm oils under two different storage conditions. J Food Sci Technol 54: 1757–1764. [CrossRef] [PubMed] [Google Scholar]
- Teklu DH, Shimelis H, Abady S. 2022. Genetic improvement in sesame (Sesamum indicum L.): progress and outlook: a review. Agronomy 12(9): 2144. [Google Scholar]
- Tinak Ekom DC, Lombeko Obe Tomo V, Neba Akongnwi C, Mahot HC. 2025. Assessing pod shattering resistance in sesame (Sesamum indicum L.): identification and evaluation of genotypic variability. BMC Agric 1(1): 8. [Google Scholar]
- Usman M, Razzaq M, Khan RAR, et al. (2022). Factors affecting postharvest losses of sesame (Sesamum indicum L.) and their mitigation strategies. Agronomy 12(10): 2470. [Google Scholar]
- Uzun B, Ülger S, Çagirgan Mİ. 2002. Comparison of determinate and indeterminate types of sesame for oil content and fatty acid composition. Turkish J Agric Forestry 26(5): 269–274. [Google Scholar]
- Uzun B, Çagirgan Mİ. 2006. Comparison of determinate and indeterminate lines of sesame for agronomic traits. Field Crops Res 96(1): 13–18. [Google Scholar]
- Wang L, Li P, Brutnell TP. 2010. Exploring plant transcriptomes using ultra high-throughput sequencing. Brief Funct Genomics 9(2): 118–128. [Google Scholar]
- Wang D, Yeats TH, Uluisik S, Rose JKC, Seymour GB. 2018. Genome-wide identification and expression profiling of the polygalacturonase (PG) and pectin methylesterase (PME) genes in grapevine (Vitis vinifera L.). Front Plant Sci 9: 1201. [Google Scholar]
- Wang K, Xie R, Ming B, Hou P, Xue J, Li S. 2021. Review of combine harvester losses for maize and influencing factors. Int J Agric Biol Eng 14(1): 1–10. [Google Scholar]
- Wang M, Huang J, Liu S, et al. 2022. Improved assembly and annotation of the sesame genome. DNA Res 29(6): dsac041. [Google Scholar]
- Wang L, Zhang Y, Li D, et al. 2023. QTL mapping in sesame (Sesamum indicum L.): a review. Ind Crops Prod 189: 116138. [Google Scholar]
- Weldemichael MY, Gebremedhn HM. 2023. Omics technologies towards sesame improvement: a review. Mol Biol Rep 50(8): 6885–6899. [Google Scholar]
- Wu H, He Q, Wang Q. 2023. Advances in rice seed shattering. Int J Mol Sci 24(10): 8889. [Google Scholar]
- Xu Y, Wang L, Liu Y, Zhang W, Wang L, Zhang Y. 2021. Fine mapping of a major pleiotropic QTL associated with sesamin and sesamolin variation in sesame (Sesamum indicum L.). Plants 10(7): 1343. [Google Scholar]
- Yol E. 2017. Inheritance of long and dense capsule characteristics in sesame. Turkish J Field Crops 22(1): 8–13. [Google Scholar]
- Zhang J, Singh AK. 2020. Genetic control and geo-climate adaptation of pod dehiscence provide novel insights into soybean domestication. G3: Genes, Genomes, Genetics 10(2): 545–554. [Google Scholar]
- Zhou R, Dossa K, Li D, et al. 2018. Genome-wide association studies of 39 seed yield-related traits in sesame (Sesamum indicum L.). Int J Mol Sci 19(9): 2794. [Google Scholar]
Cite this article as: Gholamhoseini M, Hossein Razaghi Md, Habibzadeh F, Dolatabadian A, 2026. To Shatter or Not to Shatter: Synthesising Biology and Engineering for Mechanised Harvesting of Sesame. OCL 33: 12. https://doi.org/10.1051/ocl/2025043
All Tables
TI/KE/TO Index for evaluating seed shattering resistance of Iranian sesame cultivars.
Langham’s index for evaluating seed shattering resistance in Iranian sesame cultivars.
Key ideotype traits for mechanised sesame harvesting, their inherent trade-offs, and target parameters.
Complementary morphological and physiological traits contributing to efficient direct sesame harvest.
Summary of key parameters and methods for evaluating seed loss and post-harvest quality in mechanised sesame harvesting.
Internationally recognised quality standards for graded sesame seed lots based on key physical and chemical attributes.
All Figures
 |
Fig. 1 Cross-section of a sesame capsule. Photo from Day (2000a). SU: Sutural gap, CS: Carpel wall, EN: Endocarp, EP: Epicarp, FS: False septum (pseudoseptum), L: Lumen, M: Mesocarp, P: Placenta, S: Seed, VB: Vascular bundle of the central axis. |
| In the text | |
 |
Fig. 2 Key morphological capsule traits influencing sesame seed retention and threshability. (a) Capsule dehiscence score: compares a generally shatter-resistant genotype (left) exhibiting a more complete suture split (score closer to 1), which facilitates seed release during threshing, versus a susceptible genotype (right) with a less extensive split. (b) Capsule valve opening score: illustrates differences in apical gap width (optimal score ∼ 7 allows drying while retaining seeds). (c) Membrane coverage score: shows optimal coverage (score 7), which retains seeds pre-harvest but is fragile enough for threshing. (d) Capsule compactness (cross-section): highlights reduced apical gap in resistant (left) vs. susceptible (right) capsules (optimal score 4-5 balances retention and release). (e) Seed-placenta adhesion: illustrates seed attachment within the locules (optimal score ∼ 7 withstands vibration but yields to threshing). Refer to Table 2 for detailed scoring. Scale bar in (a) = 1 cm |
| In the text | |
 |
Fig. 3 Capsule A has tip curvature after drying, making it unsuitable for mechanical harvesting; Capsule B lacks curvature but lacks shattering resistance. Capsule C combines a non-curved tip with shattering-resistant traits, making it optimal. Photo by the authors. |
| In the text | |
 |
Fig. 4 Mature sesame seeds exhibiting the visual cues of physiological maturity, including colour change and a distinct dark hilum or longitudinal line (indicated by arrows), the stage targeted for sampling in Langham’s shatter resistance assay. Photo by the authors. |
| In the text | |
 |
Fig. 5 Genetic and hormonal regulation of seed shattering resistance in sesame. The schematic illustrates key molecular and morphological determinants of shattering resistance, including: Structural gene networks (e.g., SHATTERING1 [SHAT1], INDEHISCENT [IND], and Polygalacturonase [PG] genes) controlling cell wall degradation at the abscission layer; hormonal regulation by auxin (inhibiting abscission) and gibberellin (promoting lignin deposition) and capsule morphology traits, such as dehiscence zone thickness and vascular bundle orientation, that mechanically constrain capsule opening. |
| In the text | |
 |
Fig. 6 Natural moisture variation trends in standing sesame plants (for direct harvest method, left image) versus two-stage harvested plants (right image) for whole plants, capsules, and seeds in Golestan Province, Iran. Unpublished data from the authors. |
| In the text | |
 |
Fig. 7 Height to first capsule from ground level, capsule-bearing zone length, and total plant height in Iranian sesame cultivars. Data from Siahpoosh et al. (2024) and Karimi et al. (2019). |
| In the text | |
 |
Fig. 8 Comparison of harvested sesame yield using a combine harvester with optimised settings (right) versus suboptimal configurations (left). Photo by the authors. |
| In the text | |
 |
Fig. 9 Schematic flow diagram illustrating the critical interactions between sesame capsule traits (biology) and combine harvester settings (engineering) for optimized mechanized harvesting. The diagram highlights how specific capsule characteristics necessitate adjustments in threshing, cleaning, and header units to achieve the desired outcomes (minimized loss, minimized damage, maximized purity), while emphasizing the inherent trade-offs involved in balancing shatter resistance with threshability. |
| In the text | |
 |
Fig. 10 Schematic illustration of seed shattering assessment points during mechanised sesame harvesting. (1) Natural shattering site, (2) Header unit shattering site, (3 & 4) Threshing and cleaning unit shattering sites. Source: Authors. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.




