| Issue |
OCL
Volume 33, 2026
Rapeseed / Colza
|
|
|---|---|---|
| Article Number | 13 | |
| Number of page(s) | 18 | |
| DOI | https://doi.org/10.1051/ocl/2026006 | |
| Published online | 19 March 2026 | |
Review
Deciphering canola–aphid interaction: a review on the role of hostplant nutrients and phenolic compounds☆
Décryptage des interactions colza–pucerons : revue du rôle des nutriments de la plante hôte et des composés phénoliques
1
Ministry of Science and Technology, Islamabad 44000, Pakistan
2
Department of Entomology, University of Agriculture, Faisalabad 38040, Pakistan
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
20
November
2025
Accepted:
16
February
2026
Abstract
Canola, Brassica napus L., is the top-ranked vegetable oil crop in the world, and its production is significantly hindered by aphids (mustard-oil bombs), which compromise the host plant defense. The prominent aphid species which infest the canola include Brevicoryne brassicae, Lipaphis erysimi, and Myzus persicae. They feed on tender parts of the plant and suck the cell sap that ultimately reduces the plant's vigor. Floral buds are weakened, and flowers may fall to reduce the number of potential pods, causing a significant yield loss (60%–77%). They also transmit ∼20 different plant viruses in canola. Aphids sustain their populations through a consecutive biological cycle to produce multiple generations in a single cropping season. Continuous feeding by a multitude of aphids ultimately reduces plant growth and yield attributes, i.e., plant height, number of siliques, seeds per silique, thousand-seed weight, and plant yield, significantly. Hostplant nutrient profile comprising macro- and micronutrients, i.e., nitrogen, phosphorous, potassium, calcium, magnesium, iron, sulfur, copper, and zinc, also alters. These nutrients have a dual role in aphid–canola interaction, i.e., they accomplish biological needs of aphids on one side and enhance host plant defense and yield on the other side. In addition, phenolic compounds are also important, especially myricetin, quercetin, gallic, caffeic, syringic, vanillic, ferulic, chlorogenic, m-coumaric, p-coumaric, sinapic, and cinnamic acid. Their role in plant defense and aphid reduction has been discussed. Classification, biosynthetic pathways, and the role of plant phenolics in countering aphid defense have been elaborated. Conclusively, this review delivers an inclusive framework for understanding the chemical and nutritional drivers responsible for aphid resistance and yield improvement, offering significant insights for developing pest-resilient canola cultivars.
Résumé
Le colza (Brassica napus L.) est la principale culture oléagineuse au monde, et sa production est fortement compromise par les infestations de pucerons, qui affaiblissent les mécanismes de défense de la plante hôte. Les principales espèces de pucerons infestant le colza sont Brevicoryne brassicae, Lipaphis erysimi et Myzus persicae. Ces insectes se nourrissent des parties tendres de la plante en aspirant la sève cellulaire, ce qui entraîne une réduction de la vigueur de la plante. Les boutons floraux sont affaiblis et les fleurs peuvent chuter, réduisant ainsi le nombre potentiel de siliques et provoquant des pertes de rendement significatives (60–77 %). Les pucerons sont également vecteurs d'environ vingt virus phytopathogènes chez le colza.
Les pucerons maintiennent leurs populations grâce à un cycle biologique continu, produisant plusieurs générations au cours d'une même saison culturale. Une alimentation continue de fortes populations de pucerons entraîne une diminution significative de la croissance de la plante et de ses composantes de rendement, notamment la hauteur des plantes, le nombre de siliques, le nombre de graines par silique, le poids de mille grains et le rendement global. Le profil nutritionnel de la plante hôte, comprenant les macro- et micronutriments tels que l'azote, le phosphore, le potassium, le calcium, le magnésium, le fer, le soufre, le cuivre et le zinc, est également modifié.
Ces nutriments jouent un rôle double dans l'interaction pucerons–colza : d'une part, ils satisfont les besoins biologiques des pucerons et, d'autre part, ils renforcent les défenses de la plante hôte et contribuent à l'amélioration du rendement. Par ailleurs, les composés phénoliques jouent également un rôle important, en particulier la myricétine, la quercétine, ainsi que les acides gallique, caféique, syringique, vanillique, férulique, chlorogénique, m-coumarique, p-coumarique, sinapique et cinnamique. Leur rôle dans la défense des plantes et la réduction des populations de pucerons est discuté. La classification, les voies de biosynthèse et le rôle des composés phénoliques végétaux dans les mécanismes de défense contre les pucerons sont également détaillés.
En conclusion, cette revue brosse un portrait de la compréhension des facteurs chimiques et nutritionnels responsables de la résistance aux pucerons et de l'amélioration du rendement, offrant des perspectives importantes pour le développement de cultivars de colza résistants aux ravageurs.
Key words: aphids / nutrients / phenolic compounds / phytochemistry
Mots clés : pucerons / nutriments / composés phénoliques / phytochimie
Contribution to the Topical Issue: “Rapeseed / Colza”.
© M.W. Javed Published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Highlights
This review reveals the “mustard-oil bomb” aphid impact on canola, delineating how aphid infestation disrupts nutrient and phenolics profiles and reduces yield attributes. It contributes to oilseed crop science by mapping nutrient and phenolic responses that strengthen plant defense for enhanced yield and improved pest resistance.
1 Introduction
Canola or rapeseed (Brassica napus L.) (Brassicaceae: Brassicales) is an amphidiploid dicotyledonous plant (Tsuge et al., 2020; Azhar et al., 2025). It is one of the most important oil-producing species in the genus Brassica (Esmailbegi et al., 2018; Javed et al., 2020, 2025a). Canola is thought to have evolved from other oil-producing members of the genus Brassica, i.e., B. rapa and B. oleracea. Its genome comprised of AA (2n = 20) and CC (2n = 18) from two parents (Esmailbegi et al., 2018; Azhar et al., 2025). Plants have waxy-green broad leaves, bright yellow flowers, and elongated pods, which are botanically called ‘siliques’ (Friedt et al., 2018, Javed et al., 2025b-c) (Fig. 1).
Brassica genus is famous for its oil; however, mustard oil is detrimental to human health because of high contents of glucosinolates and erucic acid (Saikia et al., 2018). Conversely, edible oil from B. napus has low levels of glucosinolates and erucic acid that make it more suitable for human consumption (Hatzig et al., 2018). Brassica napus oil ranked third in the world to fulfill global consumption (Wolko et al., 2019; Azhar et al., 2025). It is 15%–16% of vegetable oil production in the world (Confortin et al., 2019). It has high oil content (i.e., 41%–43%) (Mahmood et al., 2019), low amount of saturated fats (5%–8%) (Yousafi and Saleem, 2020), 60%–65% monounsaturated fats, and a significant amount of unsaturated fatty acids that make it important in reducing liver fat and treating cardiovascular diseases (Welter et al., 2016; Chew, 2020). It has a high smoke point, which is suitable for intensive cooking (Ajmal et al., 2018), margarine manufacturing, browning, and mixed vegetable servings (Chew, 2020).
Canola has an unachieved yield potential of 77.3% in developing countries (Ijaz et al., 2019). The yield gap exists mainly due to crop losses caused by various chewing and sucking insect pests (Shah et al., 2019). Chewing insects include cabbage butterflies (Pieris brassicae), diamondback moths (Plutella xylostella), semiloopers (Trichoplusia spp.), and mustard sawflies (Athalia lugens). Sucking insect pests are comprised of aphids (Brevicoryne brassicae, Lipaphis erysimi, and Myzus persicae) (Mpumi et al., 2020; Javed et al., 2025a–g). Canola aphids (Aphididae: Hemiptera) are the major and the most destructive insect pests of B. napus (Nasab et al., 2019; Ahmed et al., 2020; Javed et al., 2025d–g). They are hosted on a range of Brassica plants, specifically cabbage, rapeseed, mustard, cauliflower, turnip, broccoli, kale, and Brussels sprouts (Nasab et al., 2019; Razmjou et al., 2019; Javed et al., 2022). They feed on leaves, stems, and blooms to cause heavy yield losses through leaf twisting, flower falling, and pod shattering (Gill et al., 2024).
A grave attack by aphids can cause a yield loss of 60%–77% in B. napus (Aslam et al., 2011; Javed et al., 2025c). They could reduce the seed production by 9%–77% and oil content up to 11% (Mousavi, 2017). Aphids are also a vector of about 20 plant viruses, specifically cauliflower mosaic, radish mosaic, mustard mosaic, broccoli necrotic yellows, lettuce mosaic, turnip crinkle, turnip rosette, turnip yellow mosaic, and radish yellow edge virus. Through rapid reproduction, aphids defeat the plant defense, i.e., the glucosinolates-myrosinase system, for which they are also called mustard oil bombs (Ahmed et al., 2018; Ahmed et al., 2020). They sequester glucosinoates from plants and keep them separate from their body myrosinase system, and when any predator tries to feed on aphid bodies by biting, the glucosinolates get mixed with myrosinase, causing a release of toxic chemicals with a pungent odor to damage and repel the predator like a bomb (Javed et al., 2022). Aphids are also developing insecticide resistance at a rapid pace, which is making them globally important in canola cultivation (Ahmad and Akhtar, 2013; Kirkland et al., 2023).
 |
Fig 1 Canola (Brassica napus L.) plant 75 days after sowing (photo by Muhammad Wajid Javed). |
2 Biology of aphids
Canola aphids range in size from 2 to 2.5 mm. Their bodies are covered with a waxy coating in case of B. brassicae. It is difficult to distinguish B. brassicae from L. erysimi; however; L. erysimi is shorter in size (1.6 to 2.2 mm) and is without a waxy coating (Gill et al., 2024). The third species, M. persicae, is of light green to yellowish-green color with a slightly large body. Aphids reproduce both sexually (matting followed by egg laying) and asexually (parthenogenesis) (Cibils-Stewart et al., 2018) (Fig. 2a,b). Morphs that feed on B. napus are usually wingless; however, winged morphs are also produced. Cibils-Stewart et al. (2018) mentioned that under unfavorable ecological conditions, only wingless aphids are produced. Winged morphs appear only in cold winter and usually after sexual reproduction, which occurs during the time of migration (Fig. 2a).
In sexual reproduction, aphids hatch from the eggs and start to develop on primary hosts, e.g., cabbage, collards, broccoli, cauliflower, Brussels sprouts, kale, mustard, rape, kohlrabi, and turnip (Gill et al., 2024). As the contents of amino acids begin to decrease (with the maturity of plant), female forms start to develop winged morphs and begin to migrate toward secondary hosts (herbaceous plants) (Fig. 2a). Here, they develop asexually through parthenogenesis for several generations and produce wingless morphs (Awmack and Leather 2002) (Fig. 2b). There is a coincidence in the quality of primary and secondary host plants. A decrease in nitrogen in the secondary host coincides with an increase in nitrogen in the primary host. Therefore, the winged morphs are produced, which begin to migrate toward the primary host, where they will produce sexually reproducing family members (Moran, 1992). They, later on, copulate and start to lay eggs that hatch in the following year (Fig. 2a).
Biological attributes of aphids vary from species to species and crop to crop (Fig. 2c). Aslam et al. (2011) reported distinguished biological attributes of aphids on B. napus under laboratory conditions where the reproductive period of female aphids varied up to 6.25 days and fecundity (reproduction capacity) was 30.8 nymphs per female (3.85 nymphs per day). Developmental or pre-reproductive period (time required by immatures to become adults) was 12.5 days (Satar et al., 2005). Longevity duration of females was 16.3 days at 15°C temperature and 35.1 days at 10°C (Soh et al., 2018) (Fig. 2c). However, at 30°C, the longevity reduced to 9.8 days. Lifespan varied from 15.8 days (30°C) to 28.8 days (15°C). Similarly, the fecundity varied from 1.5 (30°C) to 47.1 (25°C) nymphs per female (Satar et al., 2005). Time to complete one generation was recorded as 11.3 days at 30°C and 22.6 days at 15°C, respectively. First instar needed 18.5 days to become an adult at 10°C and 5.05 days at 30°C, respectively (Soh et al., 2018). Aphids failed to become adults at higher temperatures, i.e., 35°C. Aslam et al. (2011) reported three instars of aphids, while Soh et al. (2018) mentioned four instars before reaching the adult stage (Fig. 2c).
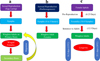 |
Fig. 2 (a) Sexual mode of reproduction, (b) asexual mode of reproduction, and (c) general biology of aphids in canola. |
3 Implications of aphids for growth and yield
During feeding, aphids inject toxic saliva into plants (Javed et al., 2020; 2022; Gill et al., 2024; Javed et al., 2025d). In response to this, growth and yield attributes begin to alter in the hostplant (Khan et al., 2015; Javed et al., 2025c,d), which is of key significance to understanding canola–aphid interaction in a dynamic ecosystem. Canola aphids cause significant losses in growth and yield attributes of B. napus by infesting the plants at various stages (Khan et al., 2015; Razaq et al., 2016; Javed et al., 2025e), which, more specifically, include flowering, blooming, and pod formation (Fig. 3). There is a significant decrease in plant height, number of siliques (pods), number of seeds per silique, thousand-seed weight, oil content, and yield per unit area in infested plants. Razaq et al. (2011) reported a yield loss of up to 75.06% in B. napus, 77.25% in B. juncea, and 81.86% in B. carinata due to aphid infestation in late-sown brassica in a field environment. Under screen house conditions, 23.65 to 62.15% yield loss was recorded in different genotypes of B. napus (Khan et al., 2015). Furthermore, the percentage recovery of oil from seeds (i.e., oil content) and its quality were also affected, with Mousavi (2017) recording an 11% reduction in oil content.
 |
Fig 3 Infested and non-infested parts of canola (Brassica napus). (a) Aphid-infested inflorescence, (b) aphid-infested pods, (c) a non-infested flower on the left and infested flowers on right side, and (d) aphid-infested pods on the left and a non-infested pod on the right side. Photo courtesy by Muhammad Wajid Javed. |
4 Nutrient responses
Plants need 14 nutrient elements for growth improvement and yield enhancement (White and Brown, 2010). These nutrients are macronutrients (required in larger amounts: 1000 to 10000 mg kg−1 of leaf dry weight). They include nitrogen (N), potassium (K), and phosphorous (P) as primary macronutrients, while calcium (Ca), magnesium (Mg), and sulfur (S) as secondary macronutrients. They are efficiently mobile inside plant's vascular area, except for Ca. Others are micronutrients (required in 0.1 to 100 mg kg−1) like iron (Fe), copper (Cu), zinc (Zn), manganese (Mn), boron (B), and chlorine (Cl) (Kirkby, 2012).
Hostplant nutrients have a dual role in aphid–canola interaction. Growth of aphids as well as plants improves when nutrient contents begin to increase (Rowntree et al., 2010). Nutrients are not only essential for plant productivity but also important for aphid performance (Bala et al., 2018). Aphids feed on the hostplant primarily to get a balanced amount of nutrients. However, feeding of aphids reduces hostplant nutrient contents. Riedell et al. (2007) reported a 25% reduction in Ca and Mg while challenged by aphids. Aphid infestation also restricts the translocation of micronutrients, e.g., Cu, Fe, and Zn (Khattab, 2007; Javed et al., 2025d). These nutrients fuel the needs of reproduction, growth, and development of aphids (Behmer, 2009). Therefore, it is important to know about the functions of various nutrients in aphids and plant bodies in order to decipher their relative role in plant defense.
4.1 Role of nutrients for aphids
Nutrients, especially N, P, and K, along with Ca, Mg, Si, and S, are among the most important ingredients to determine plant defense against insect pests. Insects usually show preference for macronutrients compared to the micronutrients, as indicated in the salt experiment (Trumper and Simpson, 1993). The same is true for aphids. Awmack and Leather (2002) say that nitrogen is one of the fundamental nutrients to determine the fecundity of aphids. Equally, potassium is one of the two important minerals in insects working to regulate cell homeostasis by Na2+/K+ pumps. At resting potential, a relatively high amount of K and a low amount of Na are present in the cell. Therefore, K is important in a number of active transport mechanisms in insects (Walter and Difonzo, 2014). Any imbalance between these two can cause paralysis or death of insects. Consequently, K is an essential nutrient in aphid hemolymph (Blow and Dougla, 2019). Being plant feeders, aphids consume a large amount of K+, and any imbalance can injure aphid biological fitness (Javed et al., 2022) (Fig. 4).
Ca is the second most important neurotransmitter in insects and controls the nervous system and functioning of muscles. Ca is a major component of eggshell, and aphids need it at the time of oviposition. Otherwise, Ca remains stored in the form of spherites in the Malpighian region (Dow, 2017). On the other hand, Mg functions as a stabilizer for polyphosphates, i.e., MgATP. Polyphosphates play their role during the synthesis of nucleic acids (DNA and RNA). Mg is also an integral component of about 300 enzymes. When the content of Mg increases in an insect's diet, Mg concentrates in the form of tubular stones in Malpighian tubules and may cause hypermagnesemia (Hofmann et al., 2010). Mg is also present in insect mitochondria to play its role in cell energetics (Dow, 2017) (Fig. 4).
Cu is an integral cofactor in a dozen enzymes; therefore, any imbalance in Cu can injure the aphid nervous system (Hwang et al., 2014). The dMTF-1 is an important metal-sensing transcriptional factor in insects to tolerate and resist excessive Cu accumulation (Chen et al., 2008). Similarly, Fe is also a cofactor for more than a hundred enzymes. It is highly toxic in comparison to other metallic ions. It is a key player in producing oxidative stress. Excessive Fe in the diet can cause hemotoxicity in aphids. Similarly, Zn is an important component in enzymatic reactions as it builds the active sites of a number of enzymes (e.g., superoxide dismutase, peptidases, carbonic anhydrase, and Zn finger nucleases—DNA binding proteins) (Carrasco-Rando et al., 2016). Both excess and deficiency of Zn are lethal for aphids (Dow, 2017).
Aphids survive better on plants with high N contents (Tao and Hunter, 2012); however, excessive feeding reduces its contents in plants. Due to the attack of B. brassicae, the uptake of nutrients is disturbed in plants. Khattab (2007) recorded a noticeable reduction in the uptake of P and Mg along with K and Ca in plants. Riedell et al. (2007) recorded a decrease in leaf N, Ca, and Mg due to infestation of different species of aphids (Schizaphis graminum, Diuraphis noxia, and Rhopalosiphum padi) on wheat and oat. Fe improves aphid reproduction, whereas Ca increases growth and decreases death rates. Ca is also an important secondary messenger and an integral component of eggshells (Dow, 2017). In addition, micronutrients (e.g., Cu, Fe, and Zn) are important in the maintenance of aphid homeostasis and intracellular symbioses (Behmer, 2009). Moreover, Zn is an important trace element to improve fecundity, growth, and physiological performance of aphids (M. persicae) (Alizamani et al., 2020) (Fig. 4). A review of the most recent studies on canola–aphid interaction with respect to nutrient impacts on aphids has also been tabulated (Table 1).
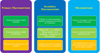 |
Fig 4 Role of primary and secondary macronutrients along with micronutrients for aphids. |
Recent studies on nutrients in aphid-canola interaction along with their major findings.
4.2 Role of nutrients for plants
In plants, nutrients are an important determinant of the yield and quality of produce. Nutrients are performing several functions in plants to specify plant growth stage where aphid infestation may cause significant damage (Fig. 5). Uchida (2000) delineated the role of nutrients in plants as follows (Fig. 6):
Canola plants take up nitrogen (N) either in the form of nitrate (NO3−) or ammonium (NH4+) ions. N is the fundamental constituent of amino acids, which are the building blocks of proteins (Okumoto and Pilot, 2011). Amino acids play their role in building cell protoplasm, which is the fundamental area of cell division. In this way, N helps in the growth and development of plants (Javed et al., 2025g). It is also an important element of enzymes in photosynthesis and respiration. Moreover, it is an integral constituent of chlorophyll and several vitamins. Nitrogen also increases the weight and quality of plant dry matter and seed proteins (Carlisle et al., 2012). Its deficiency or denaturation may cause reduction in growth due to disruption in cell division (Javed et al., 2025a). Aphid-infested, nitrogen-deficient plants show a pale yellow color, which begins from the leaf tips in canola (Khattab, 2007). Later on, chlorosis appears, leading to defoliation. In aphid-infested plants, a higher rate of nitrogen removal from apical shoots is a major cause of yield reduction (Girousse et al. 2005; Javed et al., 2025e).
Plants take phosphorous (P) in the form of orthophosphate as either HPO42–or H2PO4− ions (Shen et al., 2011). P has a major role in the energy transfer mechanism through adenosine phosphates (AMP, ADP, and ATP) during photosynthesis and respiration. It is also a vital element of DNA and RNA. It is needed in higher amounts in developing apices of shoots and roots (Shen et al., 2011). P helps in root development, flowering, and seed formation (Javed et al., 2025a). Deficiency of P slows down crop maturity and produces substandard seeds with low oil contents (Bharose et al., 2011; Javed et al., 2025c). It is a relatively mobile nutrient in plants, and its deficiency produces a dark to blue-green or purple color in plants (Uchida, 2000).
Potassium (K) is available to plants in the form of K+ ions. It is an important enzymatic activator and is vital in the growth and metabolism of canola (Myers and Gratton, 2006; Javed et al., 2025e). It helps the plants to regulate water consumption by controlling the opening and closing of stomata. During photosynthesis, K manages the electrical charge balance at the site of ATP synthesis. It also assists in translocation of sugars from roots to fruits (Safar-Noori et al., 2018). K also increases the size of the seed and improves the quality of plant produce (Javed et al., 2025c). Its deficiency can cause marginal scorching (chlorosis at the leaf margin) in older leaves. Deficiency of K can also cause stunted growth in canola (Uchida, 2000; Javed et al., 2025e). K-deficient plants have weaker stems and are susceptible to lodging and aphid attacks (Myers and Gratton, 2006; Javed et al., 2025b).
Plants take calcium (Ca) in the form of Ca2+ ions. Ca has a prominent role in cell wall synthesis, plasticity, and cell division by regulating plasma membrane permeability. It also plays an important role in the activation of enzymes during protein synthesis and carbohydrate movement (Kirkby, 2012). Ca readily links itself to sulfate, anions, and organic acids. Its combination with organic acids neutralizes them to reduce organic acid toxicity (Uchida, 2000). Ca is important in seed production (e.g., groundnut), and it improves plant yield indirectly by decreasing soil acidity, e.g., liming of soil (Estefan et al., 2013). It also works in maintaining the integrity of cell wall and regulates Na+/K+ pumps. It prevents translocation of Na+ and Cl- ions in plants to avoid salinity stress (Syeed et al., 2011). Ca is almost immobile and does not translocate in plants. Therefore, its deficiency first appears on younger parts of plants and leaf tips (Kirkby, 2012). The emerging apices of roots and leaves become brown and die, later on. Deficiency of Ca can produce weak stems. Younger leaves may stick together at the margins, which may tear apart when leaves start to grow. Floral buds may fall prematurely (Uchida, 2000; Javed et al., 2025b).
Magnesium (Mg) absorbed in the form of Mg2+ ions in plants. Mg, being the cofactor of different enzymes, activates the phosphorylation process (the fundamental step in energy generation) (Uchida, 2000). It helps in the translocation of carbohydrates in plants and maintains stability of DNA and RNA. It is a mobile nutrient and is a vital component of plant chlorophyll. Mg deficiency usually produces intervenial chlorosis that first appears on older leaves (Kirkby, 2012). B. napus may show an orange-yellow color in leaves with green veins. Mg deficiency can appear more quickly in acidic soils or soils receiving high quantities of K or Ca fertilizers (Estefan et al., 2013). Mg-deficient conditions may produce oxidative stress in plants by lowering CO2 assimilation (Uchida, 2000).
Plants take sulfur (S) in the form of sulfate SO42–ions. It is actively involved in the biosynthesis of sulfur-based amino acids (Javed et al., 2025a). Therefore, it participates in protein synthesis (Uchida, 2000). It is also important in the metabolism of vitamin B (co-enzyme A, thiamine, and biotin) (De Pascale et al., 2007). It also helps in the formation of chlorophyll, protein, seeds, nodules, oil, nutrients, glucosinolates, and phenolic contents in Brassica spp. and canola (Rehman et al., 2013; Javed et al., 2025a–d). S deficiency can produce chlorosis in younger leaves, and veins show light color. Growth of the plant reduces and maturity of the crop delays. Stems become thin and show excessive stiffness. Such stems may obstruct translocation of nutrients, and plants may lodge early (Demir and Basalma, 2018; Javed et al., 2025g). Its deficiency symptoms are similar to nitrogen and more prevalent in sandy soils with low organic matter (Estefan et al., 2013).
Plant takes copper (Cu) as Cu2+ ions. It is an integral component of several photosynthetic enzymes (Uchida, 2000). Cu is also an element of plastocyanin (a chloroplast protein in electron transport chain). It contributes to the stability and synthesis of chlorophyll and other pigments in plants. However, Cu toxicity decreases canola growth, causes distortion of new leaves, and results in loss of chlorophyll in the apical meristem (Zaheer et al., 2015; Javed et al., 2025a). Bleaching, defoliation, and twig dieback may occur later on. Male and female reproductive organs may become sterile (Khandekar and Leisner, 2011).
Plants take iron (Fe) either in the form of Fe2+ or Fe3+ ions. It is an important part of heme enzymes (during photosynthesis). Fe is also an integral part of several enzymes that participate in respiration, i.e., cytochrome oxidase and catalase (Uchida, 2000). Fe, as a component of ferredoxin, is required in the reduction of sulfate and nitrates. It also contributes to the metabolism of proteins (Pavlovic et al., 2013). It is also involved in the biosynthesis of chlorophyll molecules. Therefore, Fe deficiency can cause interdental chlorosis, which is more common in alkaline or excessively limed soils. Newly emerging leaves may appear white (Uchida, 2000).
Plants uptake zinc (Zn) in the form of Zn2+ ions (Roy and Ghosh, 2020). Zn has an important role in the formation of tryptophan (a player in the production of indole acetic acid in plants). It is important in several metabolic processes, as it is a part of different metal-containing enzymes, e.g., dehydrogenases (Uchida, 2000). It activates carbonic anhydrase enzyme, specifically. Zn is also vital in the production of protein and RNA, including Zn-finger transcription factors (Carrasco-Rando et al., 2016). In B. napus, Zn deficiency can produce stunted plants with inter-venial chlorosis on lower, older leaves. Sometimes, chlorotic tissues may drop out from the leaf area. This may alter plant photosynthesis and growth (Uchida, 2000).
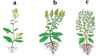 |
Fig 5 Nutrient flux-mediated growth stages of canola where aphids can cause significant damage. (a) Flowering, (b) blooming, and (c) pod formation. |
4.3 Role of nutrients in plant defense against aphids
Any alteration in nutrient contents in plants is strongly linked to plant defense and performance of aphids. Tao and Hunter (2012) showed that feeding of aphids on nutritionally deficient plants reduces aphid body mass. High activity of aphids was noted on plants with higher nutrient contents, particularly nitrogen (Garratt et al., 2010a,b). Nitrogen is one of the most important nutrients to improve the aphid performance. Most of the studies reported an improved development of herbivores under an excessive supply of nitrogen (Lu et al., 2007). A few studies mentioned that nitrogen negatively affects the development of aphids on canola when applied at moderate rates (Javed et al., 2025a, e). Nitrogen affects the growth of aphids by regulating the availability of amino acids (Javed et al., 2022). Excessive use of nitrogen was found to reduce the contents of lignin in plants, which is the fundamental constituent of resistance against aphids (Torres-Olivar et al., 2014). An adequate supply of nitrogen helps the plants to synthesize physical defense compounds, e.g., lignin and suberin, as a potent tool of aphid resistance (Wang et al., 2020).
Conversely, phosphorus (P) is not a limiting factor for insect growth as nitrogen is. Due to its role in the synthesis of ATP, enzymes, and nucleic acids, phosphorus plays a diverse role in plant defense, especially against hemipterans (Singh and Sood, 2017). Equally, potassium (K) defends the plants by modulating biochemical contents, specifically amino acids, silica, and carbohydrates in plants (Imas, 2013). K, again, is an important regulator of Na/K pump to maintain homeostasis in aphids, and its imbalance affects aphids negatively (Dow, 2017; Singh and Sood, 2017).
Due to injury of aphids, the particular type of proteins called forisome accumulates across the wounded sieve tubes to restrict the loss of phloem (Will et al., 2007). This plugging is mediated by the influx of Ca2+ ions. Aphids adopt an alternate mechanism for continuous flow of sap in their mouths. They increased the transcript levels of a number of Ca-binding proteins, e.g., calmodulin-binding proteins, calcium-dependent protein kinases, pinoid-binding protein, calcium-transporting ATPases, and calcium-binding calreticulin (Giordanengo et al., 2010). Induction of Ca-binding proteins produces senescence in plants. Brevicoryne brassicae was found to induce senescence-associated proteins in A. thaliana (Will et al., 2007). Being an important component of insect eggshells, any imbalance in Ca can significantly affect the new offspring of aphids (Dow, 2017).
In a similar way, magnesium (Mg) also defends against insects, as it is an important stabilizer of ATPs and ADPs. An increase in the contents of Mg during feeding increases the chances of tubular stones in insects (Wessing and Zierold, 1992). Also, iron (Fe) is a cofactor of several enzymes in insects. Fe can cause damage to aphids by hemotoxicity (Graca-Souza et al., 2006). Parallel to these, copper (Cu) defends the plants by damaging the nervous system of insects (Hwang et al., 2014). It is also a part of several enzymes as a cofactor. Similarly, zinc (Zn) is also a critical element, and it forms the active sites of several enzymes. Excess of Zn can generate hard concretions, e.g., Zn-stones in aphids and other insect pests (Killilea et al., 2015).
5 Phenolic responses
Phenolics are the allelochemicals that perform various functions to defend the plants against aphids and other pests. Phenolic compounds affect the food intake, disturb the enzymatic catalysis, and delay aphid growth (Czerniewicz et al., 2017; Javed et al., 2025c). Vuolo et al. (2019) defined the phenolic compounds as one of the important anti-oxidative metabolites that are biosynthesized through the phenylpropanoid or shikimate pathway. Chemically they are composed of a benzene ring with one or several hydroxyl subunits (Tsao, 2010). They are usually derived from phenylalanine, which, in turn, is produced by the activity of phenylalanine ammonia-lyase (PAL) enzymes (Tajik et al., 2019). This enzyme is induced particularly from the elicitation of phytohormones (e.g., jasmonic acid), which are produced under octadecanoid pathways. Later on, they transfer the amino acids to the site of production of the secondary metabolites.
Several studies have shown the defense role of phenolic compounds against insects or pathogens (Khoshfarman-Borji et al., 2020; Javed et al., 2025c–e). Besides plant defense, these chemicals provide color, scent, and other tangible properties to plants. They also contribute in indirect plant defense through alternation in host plant quality to restrain aphid development and modify host plant preference (Khoshfarman-Borji et al., 2020; Javed et al., 2022, 2025b–d). Kumar et al. (2017) showed that phenolic compounds were inversely related to aphid preference. Phenolic compounds are also the best antioxidants to relieve plants from oxidative stress (Tajik et al., 2019).
5.1 Classification of phenolic compounds
Jacobo-Velázquez and Cisneros-Zevallos (2020) classified the phenolic compounds on the basis of hydroxy or methoxy subunits. Phenolic compounds have one hydroxy (4-hydroxybenzoic acid and p-coumaric acid), one hydroxy and one methoxy (vanillic acid and ferulic acid), one hydroxy and two methoxy groups (syringic acid and sinapic acid), and two hydroxy subunits (protocatechuic acid and caffeic acid). In Brassica, sinapic acid is the most abundant phenolic compound (Williamson and Clifford, 2010). Phenolic acids can be either hydroxy cinnamic acid (C6-C3) or hydroxy benzoic acid (C6-C1), with at least one benzene ring in their structure (Williamson and Clifford, 2010). Chrzanowski (2012) categorized phenolic acids on the basis of the number of carbon atoms (length of the side chain), e.g., benzoic, cinnamic, and phenylacetic acid.
Hydroxycinnamic acids (C6–C3) exist either in cis or trans formation with double bonds. They exist in the form of hydroxy acid condensates, mono/disaccharides (producing esters), alcohols, or amides. While hydroxybenzoic acid (C6–C1) usually comprised of ellagic, gallic, salicylic, protocatechuic, 4-hydroxybenzoic, gentisic, syringic, vanillic, and hexa-hydroxy-diphenic acids (dilactone and ellagic acid). They occur in conjugate forms (Williamson and Clifford, 2010). Formation of hydroxybenzoic acid is rather a complex process. They may be the direct product of the shikimate pathway or produced from the derivatives of cinnamic acid (Averesch and Krömer, 2018). The basic framework of vanillic acid derives from ferulic acid, whereas that of syringic acid derives from sinapic acid. Caffeic acid or p-hydroxybenzoic acid produces protocatechuic acid. Hydroxybenzoic acids also produce vanillic, gallic, syringic, and protocatechuic acids (Spilioti et al., 2014).
Parallel to these, flavonoids are the largest group of phenolic compounds having low molecular weight, carrying 15 carbons (C6–C3–C6). They have two benzene rings (A and B), which are held together by a C3 linkage. Ring A is derived from acetate via the malonate pathway, whereas ring B is derived from phenylalanine by the shikimate pathway (Williamson and Clifford, 2010). In addition, the third ring, called ring C, is responsible for producing varieties in flavonoids, e.g., anthocyanidin, flavonols, flavanols (catechin), flavanones, flavones, isoflavones, and flavanonols.
Flavonoids are among the most important groups. They exist as glycosides with relatively stable bonds with sugars, and hence, are less bioavailable. They are yellow or red in color due to the presence of chromophores, which produce different colors in seeds, flowers, and fruits. Flavonols and flavones are red in color, whereas flavanones are either brownish or colorless (Brouillard and Dangles, 2017). They exit as polyphenols (Brouillard and Dangles, 2017). They contain more than one aromatic ring, mostly C15 (Iwashina, 2003). Flavonoids destroy free radicals, and therefore, they are important against herbivore-induced oxidative stress (Brouillard and Dangles, 2017). Around 5000 flavonoids are present in plants, and only a few of them are responsible for plant defense (Iwashina, 2003). Khan et al. (2011) and Javed et al. (2025c) found that flavonoids, especially kaempferol, quercetin, and myricetin, were indispensably reduced in B. napus due to B. brassicae infestation. Xiao et al. (2019) also described a significant manifestation of flavonoids in response to herbivores in Brassica. They found quercetin as a major flavonoid that exist as glycosides called rutin (Ateyyat et al., 2012).
Roughly 70% of plants contain flavonoids. Other groups are anthocyanidins, proanthocyanidins, leucoanthocyanidins, flavonol, flavones, and flavan (Iwashina, 2003). Flavonols (myricetin, quercetin, kaempherol, isorhamnetin, and rutin) are among the most important class of flavonoids, which are responsible for pest resistance (Samanta et al., 2011). In flavonoids, the occurrence of two OH groups contributes to antioxidant activity, while the third OH group reduces this activity (Qayyum et al., 2016). Table 2 describes categories, specific compounds, and the significance of various phenolic compounds that exist in canola (B. napus).
Categories, sub-types, specific compounds, and the significance of major classes of phenolic compounds in canola.
5.2 Biosynthesis of phenolic compounds
Phenolic compounds are produced due to the transformation of benzene ring-based amino acids, e.g., phenylalanine and tyrosine (Chrzanowski, 2020). Anaerobic removal of the ammonia group from phenylalanine and tyrosine in the presence of L-phenylalanine ammonia-lyase (PAL) and tyrosine ammonia lyase (TAL) produces trans-cinnamic acid and p-coumaric acid, respectively (Czerniewicz et al., 2017). Later on, condensation of three acetate subunits with p-coumaroyl-CoA, catalyzed by chalcone synthase, produces various classes of flavonoids (e.g., anthocyanins, proanthocyanidins, flavones, flavandiols, flavonols, and isoflavonoids) (Ferrer et al., 2008). Products of phenylpropanoids produce ferulic acid (dehydration), caffeic acid (hydroxylation), and p-coumaric acid (methylation). Both free and complex formations of cinnamic acids exist in the form of depsides or glycosides. They are present in cell nucleus, cell wall, and vacuole (Czerniewicz et al., 2017).
Hydroxycinnamic acid and hydroxybenzoic acid are the derivatives of non-phenolic cinnamic and benzoic acid, which are biosynthesized through the shikimate pathway from L-phenylalanine and L-tyrosine, respectively (Williamson and Clifford, 2010). Initially, L-phenylalanine and L-tyrosine undergo deamination to produce either cinnamic or p-coumaric acid. Later on, their benzene rings combine with hydroxyl (OH) groups (i.e., hydroxylation) and methyl (CH3) groups (i.e., methylation) to produce ferulic and caffeic acid, respectively. Occasionally, the branched chain of cinnamic acid may be degraded to produce benzoic acid, which is hydroxylated and methylated to generate protocatechuic acid and p-hydroxybenzoic acid (Vuolo et al., 2019).
Another explanation of phenolic compound biosynthesis is based on the production of cinnamic acid from L-phenylalanine (under the action of PAL enzymes). This is the very first stage to produce phenolic compounds. Later on, cinnamic acid is changed into p-coumaric acid under the action of cinnamic acid 4-hydroxylase (CAH). Then p-coumaric acid is converted into caffeic acid and p-hydroxybenzoic acid (both are the derivatives of hydroxybenzoic acid). Caffeic acid is methylated to form ferulic acid. Ferulic acid is then methylated to produce sinapic acid. Later on, o-coumaric acid is also formed from cinnamic acid to produce salicylic acid. Salicylic acid is finally transformed into gentisic acid that, in turn, changes into o-pyrocatechuic acid (Fig. 7).
 |
Fig 6 Categorical role of macronutrients (primary and secondary) and micronutrients for canola. |
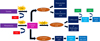 |
Fig 7 Biosynthesis of hydroxybenzoic and hydroxycinnamic acid from amino acids tyrosine and phenylalanine, using tyrosine ammonia lyase (TAL), L-phenylalanine ammonia-lyase (PAL), and cinnamic acid 4-hydroxylase (CAH) enzymes. NH3 and CH3 show deamination and methylation, respectively. Figure courtesy by Muhammad Wajid Javed. |
5.3 Mode of action of phenolic compounds against aphids
Phenolic acid and polyphenol oxidases (PPO) are naturally present in plants in isolated organelles. When the tissue is damaged due to aphid puncture, the phenolics and their detoxifying enzymes encounter each other to elicit a potent phenolic defense response (Altunkaya and Gökmen, 2009). Brassica phenolics produce hostplant defense against aphids through antibiosis, antixenosis, and anti-symbiosis (Javed et al., 2020, 2025e). Chrzanowski et al. (2012) and Javed et al. (2025c) described the antibiotic mechanism of phenolic compounds against aphids through reduction in development, survival, and reproduction fitness. Infestation of aphids increases the synthesis of amino acid peptides (e.g., systemin) to elicit plant lipase in cell membrane to liberate linolenic acid. The linolenic acid induces jasmonic acid pathway to produce proteinase inhibitor, peroxidase, and polyphenol oxidase (Vuolo et al., 2019).
These oxidizing enzymes act on plant phenolics to form quinones and reactive oxygen species. Quinones, when ingested by the aphids, work as anti-nutritional proteins to reduce nutrient intake and food digestion (Dampc et al., 2020; Javed et al., 2025b, f). The proteinase inhibitors attach themselves to aphid digestive enzymes in the midgut, therefore affecting food digestion. Phenolics may also inflict injuires to aphid midgut by the mechanism of oxidative stress. They freely bind with thiols inside midgut epithelium to decrease the availability of non-protein thiols and vitamin C (ascorbic acid).
Similarly, flavonoid, e.g., quercetin, interferes in the insect molting by reducing the expression of ecdysone receptor (EcR) genes (Oberdörster et al., 2001). Besides these, flavonoids and phenolic acids (vanillic, ferulic, and 4-hydroxybenzoic acids) have been recorded to suppress the acetylcholinesterase enzyme, which is responsible for molting in rice weevils (Maazoun et al., 2019). Flavonoids, e.g., taxifolin is documented to reduce glutathione S-transferase expression (insecticide detoxifying enzyme in insects) (Wang et al., 2016).
Phenolic compounds also cause antixenosis. They make the plant a less preferable host and reduce food palatability (Chrzanowski et al., 2012). Dreyer and Jones (1981) reported dihydrochalcones (flavonoids), as an effective deterrent for aphids (e.g., S. graminum and M. persicae). Phenolics may change the plant morphological attributes (wax, hairiness, and color) or discharge deterrent volatile compounds (Vendramim and Guzzo, 2012), which ultimately result in reduced host colonization. However, if the aphids have still been able to colonize the plants, phenolics may trigger a hypersensitive response using peroxidase enzymes to lignify plant tissues. This lignification appears as a strong physical barrier against aphid stylets. In addition, peroxidase transforms chlorogenic acid to chloroquinone, which attaches to the amino acids to decrease their availability (Appel, 2017).
Anti-symbiosis is another mechanism of phenolic defense to reduce aphid populations (Czerniewicz et al., 2017; Javed et al., 2025a–e). Czerniewicz et al. (2017) demonstrated that an increase in the contents of various phenolics negatively correlated with the population of aphids (S. avenae) in wheat. Phenolics may affect pre-reduction time, fertility, and population growth of aphids (Gantner et al., 2019). In the same way, flavonoids (quercetin and myricetin) cause endocrine toxicity in aphids. Both quercetin and myricetin, in the presence of Fe3+, enhances the production of OH radicals from hydrogen peroxide at pH 4.7. Quercetin and myricetin may also cause DNA damage in aphids (Ateyyat et al., 2012). Quercetin causes DNA damage through the production of hydrogen peroxide (Sosa et al., 2000).
Therefore, to counter the phenolic defense, aphids detoxify the phenolic compounds and transform them into non-toxic derivatives using salivary sheath enzymes (peroxidases, phenoloxidases, and pectinases) (Cherqui and Tjallingii, 2000). In short, plant triggers the phenolic defense in response to aphid feeding. Simultaneously, aphid tries to counter the defense by synthesizing peroxidase and polyphenol oxidase to oxidize phenolics into less toxic compounds (Xiao et al., 2019; Dampc et al., 2020). Table 3 highlights major studies on recent findings in aphid-phenolic interactions in canola.
Recent studies on aphid-phenolic interactions in canola.
5.4 Functions of plant phenolic compounds
The mentioned functions of phenolic compounds are referred from PubChem (https://pubchem.ncbi.nlm.nih.gov/).
Myricetin is an efficient inhibitor of cyclooxygenase-1, which is responsible for preventing neoplasm or tumor. It is an important plant metabolite, an antioxidant, a hypoglycemic agent, and a food component. Quercetin is an anti-cancer agent, a chief bioflavonoid in human food, an antioxidant and antibacterial agent. It prohibits cell cycle at G1 phase and efficiently inhibits heat shock proteins. This also inhibits lipoxygenase and cyclooxygenase pathways to restrict inflammatory agents. Quercetin also serves as an important scavenger of radicals.
Gallic acid is an astringent (constricts the skin), an antioxidant, an inhibitor of cyclooxygenase-2, an important plant metabolite, an antineoplastic agent, a human xenobiotic, and an inducer of apoptosis. Syringic acid is an important plant metabolite, which is derived from gallic acid. Vanillic acid is also an important plant metabolite and flavoring agent. It is an intermediary product of the biotransformation of ferulic acid to vanillin.
Caffeic acid is an orally bioavailable plant metabolite with anti-oxidative, anti-inflammatory, and anti-cancer properties. It averts free radical-mediated DNA damage. Its trans form is more abundant, but cis structure is also present in plants. It is an inhibitor of lipoxygenase and glutathione transferase. Chlorogenic acid is a product of trans-caffeic acid and quinic acid. This is an intermediate metabolic product during the synthesis of lignin. It is actually a tannin.
Ferulic acid also existed in cis and trans structures. It is an antioxidant, a plant metabolite, an anti-inflammatory agent, an apoptosis inhibitor, a human xenobiotic, and a MALDI (matrix-assisted laser desorption/ionization) matrix material. Coumaric acid is present in ortho, meta, and para formations. It is also an anti-inflammatory, antimicrobial, anticancer, and antioxidant agent. It reduces peroxidation of lipids with antioxidant and antimicrobial properties. Sinapic acid is also an important antibacterial, anti-inflammatory, anti-mutagenic, anti-glycemic, anticancer, and neuroprotective agent.
5.5 Role of phenolic compounds in plant defense against aphid
Studies have described the role of phenolic defense during aphid feeding, which is regulated by PAL enzymes (Smith and Boyko, 2007). Phenolic compounds that work against aphids may comprise myricetin and quercetin (flavonoid), gallic acid, syringic acid, and vanillic acid (hydroxybenzoic acid), along with caffeic acid, chlorogenic acid, ferulic acid, m-coumaric acid, p-coumaric acid, sinapic acid, and trans-4-hydroxy-3-methoxy cinnamic acid (hydroxycinnamic acid) (Chrzanowski and Leszczyński, 2008; Czerniewicz et al., 2017; Gantner et al., 2019). Ciepiela and Chrzanowski (2001) recorded higher contents of ferulic acid in aphid-resistant winter triticale. Aphid biological fitness was also affected negatively due to different phenolic acids.
Here, p-coumaric acid reduced the number of S. avenae nymphs (Chrzanowski, 2012). Chrzanowski (2012) also recorded a noticeable decrease in fecundity and population growth when S. avenae fed on caffeic acid-treated plants. Cabrera et al. (1995) also noticed that feeding of aphids could activate the plant phenolic defense and could increase the contents of caffeic acid to reduce the survival of aphids. Gantner et al. (2019) also reported a smaller number of aphids (Myzocallis coryl) on hazel cultivars having higher contents of gallic, chlorogenic, and caffeic acid.
Chrzanowski and Leszczyński (2008) also showed that wheat plants with higher levels of ferulic and o-coumaric acid usually had lower populations of aphids. However, Gantner et al. (2019) presented a contradictory finding and showed that higher ferulic acid may not make the plant resistant to aphids. Czerniewicz et al. (2017) showed that the contents of caffeic, o-coumaric, p-coumaric, and chlorogenic acid increased in response to aphid feeding, but levels of ferulic acid remained unchanged. A few important phenolic acids, e.g., chlorogenic acid, p-hydroxybenzoic acid, and (+)-catechin influenced the migratory flight of aphids (R. padi) from bird cherry-oats toward triticale and orchard grasses (Czerniewicz et al., 2011).
In flavonoids, quercetin and its derived form (rutin) are the most important group of flavonols that work against aphids. Tosh et al. (2003) reported a number of negative effects of quercetin on insect pests. Ateyyat et al. (2012) also found a significant decline in aphid population (Eriosoma lanigerum) in response to quercetin, rutin (a derivate of quercetin), and naringin. Sosa et al. (2000) found quercetin to reduce larval growth in Tenebrio molitor.
Łukasik and Goławska (2013) showed that phenolics (quercetin) also prolonged the pre-reproductive period and developmental time of aphids (S. avenae and R. padi). Higher mortalities were reported on plants having higher contents of phenolics. On the other hand, lower contents of quercetin reduced the ingestion, while higher contents blocked the feeding of aphids. Łukasik and Goławska (2013) also demonstrated negative effects of phenolics (luteolin and genistein) on feeding of aphids (Acyrtosiphon pisum). Similarly, Łukasik et al. (2017) showed the negative impacts of quercetin on aphids (A. pisum) on Fabaceous plants.
6 Prospects and applications of the nutrients and phenolics for canola development
The future of protecting canola crops relies on leveraging interaction between plant nutrition and phenolic chemistry. Instead of depending solely on heavy sprays of pesticides, researchers are looking at how we can fuel a plant's internal security system. By fine-tuning the nutrients we provide—specifically sulfur, potassium, and silicon—we aren't just making the plant grow faster; we are effectively turning the switch on for the plant's ability to produce phenolic compounds. These phenolics, which include substances like ferulic, chlorogenic, caffeic, coumaric, and sinapic acids, act as a multi-layered defense. They lignified the plant's cell walls, making it physically harder for aphids to pierce the tissue, and they turned the plant's sap into an indigestible, bitter, unappealing food that stunts the pests' ability to survive.
The real prospect here is moving toward a “bottom-up” defense-mediated strategy. By using metabolic engineering or precision nutrient application, a canola plant's defense could be optimized against aphids. This shift is not just about plant health; it is about a more sustainable way toward canola production. When a plant can defend itself through activation of its own built-in defense, we reduce our dependence on synthetic chemicals, protecting the environment while ensuring that canola remains a high-yielding crop for the future.
7 Conclusion
Both aphids and canola have developed an intricate pattern of relation that is dynamic in a continuously evolving environment. Aphids feed on canola plants and cause severe damage. However, hostplant nutrients and phenolic compounds in canola play a vital role for plant defense against aphids. Aphids require nutrients for biochemical and biological needs, while plants require them for growth, yield, and sustainable vigor. Both macro- and micronutrients are essential for aphids as well as canola growth, having different roles and functions in both organisms. On similar lines, the role of phenolic compounds is also pivotal in aphid–canola interaction, where phenolic compounds play an interactive role of defense, plant vigor improvement, and aphid management. Instead of a single line of defense, phenolic compounds target aphids with a multilayered defense: they strengthen the plant's tissues to make them physically harder to feed, they make the sap into a less digestible component, and they even release plant volatiles that produce an antixenotic response in aphids. By focusing on these, we can breed “smarter” canola varieties that are naturally resilient and consume less pesticides and provide crop produce.
Acknowledgments
The author acknowledges the critical comments and review by Dr. Lewis J. Wilson, CSIRO, Australia to improve the quality of this manuscript.
Funding
No funding was received for this publication.
Conflicts of interest
The author has no conflict of interest to declare related to this publication.
Author contribution statement
Muhammad Wajid Javed conceptualized, collected the data, wrote, and reviewed the manuscript.
References
- Abbas D, Liaqat U, Ali H, et al. 2026. Management of canola aphids (Hemiptera: Aphididae) using potassium silicate (K2SiO3) in integration with Chrysoperla carnea Stephens. J. Plant Dis. Prot. 133: 8. [Google Scholar]
- Ahmad M, Akhtar S. 2013. Development of insecticide resistance in field populations of Brevicoryne brassicae (Hemiptera: Aphididae) in Pakistan. J. Econ. Entomol. 106: 954-958. [Google Scholar]
- Ahmed M, Qin P, Ji M, An R, Guo H, Shafi J. 2020. Spinasterol, 22, 23-Dihydrospinasterol and Fernenol from Citrullus Colocynthis L. with aphicidal activity against cabbage aphid Brevicoryne brassicae L. Molecules 25: 2184-2191. [Google Scholar]
- Ahmed N, Darshanee HLC, Fu WY, Hu XS, Fan Y, Liu TX. 2018. Resistance of seven cabbage cultivars to green peach aphid (Hemiptera: Aphididae). J. Econ. Entomol. 111: 909-916. [Google Scholar]
- Ajmal M, Nadeem M, Batool M, Khan IT. 2018. Probable ingredients for trans free margarine with omega-3 fatty acids. Biol. Sci-PJSIR. 61: 182-186. [Google Scholar]
- Alizamani T, Shakarami J, Mardani-Talaee M, Zibaee A, Serrão JE. 2020. Direct interaction between micronutrients and bell pepper (Capsicum annum L.), to affect fitness of Myzus persicae (Sulzer). J. Plant Prot. Res. 60: 253-262. [Google Scholar]
- Altunkaya A, Gökmen V. 2009. Effect of various anti-browning agents on phenolic compounds profile of fresh lettuce (L. sativa). Food Chem. 117: 122-126. [Google Scholar]
- Appel HM. 2017. The chewing herbivore gut lumen: Physicochemical conditions and their impact on plant nutrients, allelochemicals, and insect pathogens. In: E.A. Bernays, ed. Insect-Plant Interactions. CRC Press. Boca Raton. Vol. 5. pp. 209-224. [Google Scholar]
- Aslam M, Amer M, Shad SA. 2011. Insect pest status of aphids on oilseed brassica crops and need for chemical control. Crop Environ. 2: 60-63. [Google Scholar]
- Ateyyat M, Abu-Romman S, Abu-Darwish M, Ghabeish I. 2012. Impact of flavonoids against woolly apple aphid, Eriosoma lanigerum (Hausmann) and its sole parasitoid, Aphelinus mali (Hald.). J. Agric. Sci. 4: 227-236. [Google Scholar]
- Averesch NJ, Krömer JO. 2018. Metabolic engineering of the shikimate pathway for production of aromatics and derived compounds-present and future strain construction strategies. Front. Bioeng. Biotechnol. 6: e32. [Google Scholar]
- Awmack CS, Leather SR. 2002. Host plant quality and fecundity in herbivorous insects. Ann. Rev. Entomol. 47: 817-844. [Google Scholar]
- Azhar M, Cahill DM, Khan GA. 2025. A brief history of canola genetic gains: from classical breeding to genome editing. Physiol. Plant 177:e70644. [Google Scholar]
- Bala K, Sood A, Pathania VS, Thakur S. 2018. Effect of plant nutrition in insect pest management: a review. J. Pharm. Phytochem. 7: 2737-2742. [Google Scholar]
- Behmer ST. 2009. Insect herbivore nutrient regulation. Ann. Rev. Entomol. 54: 165-187. [Google Scholar]
- Bharose R, Chandra S, Thomas T, Dhan D. 2011. Effect of different levels of phosphorus and sulphur on yield and availability of N, P, K, protein and oil content in Toria (Brassica spp.). J. Agric. Biol. Sci. 6: 31-33. [Google Scholar]
- Blow F, Douglas AE. 2019. The hemolymph microbiome of insects. J. Insect Physiol. 115: 33-39. [Google Scholar]
- Brouillard R, Dangles O. 2017. Flavonoids and flower color. In: J.B. Harborne, ed. The flavonoids. New York: Routledge. pp. 565-588. [Google Scholar]
- Cabrera H, Munoz O, Zuniga G, Corcuera L, Argandona V. 1995. Changes in ferulic acid and lipid content in aphid-infested barley. Phytochemistry 39: 1023-1026. [Google Scholar]
- Carlisle E, Myers SS, Raboy V, Bloom AJ. 2012. The effects of inorganic nitrogen form and CO2 concentration on wheat yield and nutrient accumulation and distribution. Front. Plant Sci. 3: e195. [Google Scholar]
- Carrasco-Rando M, Atienza-Manuel A, Martín P, Burke R, Ruiz-Gómez M. 2016. Fear-of-intimacy-mediated zinc transport controls the function of zinc-finger transcription factors involved in myogenesis. Development 143: 1948-1957. [Google Scholar]
- Chen X, Hua H, Balamurugan K, et al. 2008. Copper sensing function of Drosophila metal-responsive transcription factor-1 is mediated by a tetra-nuclear Cu (I) cluster. Nucleic Acids Res. 36: 3128-3138. [Google Scholar]
- Cherqui A, Tjallingii WF. 2000. Salivary proteins of aphids, a pilot study on identification, separation and immunolocalisation. J. Insect Physiol. 46: 1177-1186. [Google Scholar]
- Chew SC. 2020. Cold-pressed rapeseed (Brassica napus) oil: Chemistry and functionality. Food Res. Int. 131: e108997. [Google Scholar]
- Chrzanowski G, Leszczyński B. 2008. Induced accumulation of phenolic acids in winter triticale (Triticosecale Wittm.) under insects feeding. Herba Pol. 54: 33-40. [Google Scholar]
- Chrzanowski G, Leszczyński B, Czerniewicz P, et al. 2012. Effect of phenolic acids from black currant, sour cherry and walnut on grain aphid (Sitobion avenae F.) development. Crop Prot. 35: 71-77. [Google Scholar]
- Cibils-Stewart X, Nechols J, Giles K, McCornack BP. 2018. Feeding location of aphid prey affects life history traits of a native predator. Entomol. Exp. Appl. 156: 149-159. [Google Scholar]
- Ciepiela AP, Chrzanowski G. 2001. Accumulation of ferulic, gallic, o-coumaric and salicylic acid in winter triticale of different resistance to grain aphid (Sitobion avenae (F.), Homoptera: Aphididiade). Monograph of aphids and other homopterous insects (Ed. 8th). Siedlce : PAS. pp. 205-211. [Google Scholar]
- Confortin TC, Todero I, Luft L, et al. 2019. Oil yields, protein contents, and cost of manufacturing of oil obtained from different hybrids and sowing dates of canola. J. Environ. Chem. Eng. 7: 10-19. [Google Scholar]
- Czerniewicz P, Leszczynski B, Chrzanowski G, Sempruch C, Sytykiewicz H. 2011. Effects of host plant phenolics on spring migration of bird cherry-oat aphid (Rhopalosiphum padi L.). Allelopathy J. 27: 309-316. [Google Scholar]
- Czerniewicz P, Sytykiewicz H, Durak R, Borowiak-Sobkowiak B, Chrzanowski G. 2017. Role of phenolic compounds during antioxidative responses of winter triticale to aphid and beetle attack. Plant Physiol. Biochem. 118: 529-540. [Google Scholar]
- Dampc J, Kula-Maximenko M, Molon M, Durak R. 2020. Enzymatic defense response of apple aphid Aphis pomi to increased temperature. Insects 11: e436. [Google Scholar]
- De Pascale S, Maggio A, Pernice R, Fogliano V, Barbieri G. 2007. Sulphur fertilization may improve the nutritional value of Brassica rapa L. subsp sylvestris. Eu. J. Agron. 26: 418-424. [Google Scholar]
- Dehghan A, Rounagh-Ardakani H, Mohammadzadeh A, Mohammadzadeh M, Mohammadzadeh M, Borzoui E. 2023. Induction of resistance, enzyme activity, and phytochemicals in canola plants treated with abscisic acid elevated based on nutrient availability: a case study on Brevicoryne brassicae L. (Hemiptera: Aphididae). J. Insect Sci. 23: 17. [Google Scholar]
- Demir I, Basalma D. 2018. Response of different level of nitrogen and sulphur doses on oil yield and seed nutrients content of sunflower (Helianthus annuus L.). Fr. Environ. Bull. 27: 6337-6342. [Google Scholar]
- Dow JA. 2017. The essential roles of metal ions in insect homeostasis and physiology. Cur. Op. Insect Sci. 23: 43-50. [Google Scholar]
- Dreyer DL, Jones KC. 1981. Feeding deterrency of flavonoids and related phenolics towards Schizaphis graminum and Myzus persicae: Aphid feeding deterrents in wheat. Phytochemistry 20: 2489-2493. [Google Scholar]
- Esmailbegi S, Al-Shehbaz IA, Pouch M, et al. 2018. Phylogeny and systematics of the tribe Thlaspideae (Brassicaceae) and the recognition of two new genera. Taxon 67: 324-340. [Google Scholar]
- Estefan G, Sommer R, Ryan J. 2013. Methods of soil, plant, and water analysis: A manual for the West Asia and North Africa region, 3rd ed. Beirut, Lebanon: International Center for Agricultural Research in the Dry Areas (ICARDA).. [Google Scholar]
- Farhan M, Pan J, Zhao J, Yang H, Zhang S. 2025. Aphid adaptation to plant secondary metabolites: adaptive mechanism of resistance evolution and future prospects. Hort. Res. 13(1) 269. [Google Scholar]
- Farhan M, Pan J, Hussain H, et al. 2024. Aphid-resistant plant secondary metabolites: types, insecticidal mechanisms, and prospects for utilization. Plants 13: 2332. [Google Scholar]
- Ferrer JL, Austin M, Stewart C, Noel J. 2008. Structure and function of enzymes involved in the biosynthesis of phenylpropanoids. Plant Physiol. Biochem. 46: 356-370. [Google Scholar]
- Friedt W, Tu J, Fu T. 2018. Academic and economic importance of Brassica napus rapeseed. In: S. Liu, R. Snowdon and B. Chalhoub, eds. The Brassica napus genome. Springer Nature Switzerland. pp. 1-20. [Google Scholar]
- Gantner M, Najda A, Piesik D. 2019. Effect of phenolic acid content on acceptance of hazel cultivars by filbert aphid. Plant Prot. Sci. 55: 116-122. [Google Scholar]
- Garratt MP, Wright DJ, Leather SR. 2010a. The effects of organic and conventional fertilizers on cereal aphids and their natural enemies. Agric. Forest Entomol. 12: 307-318. [Google Scholar]
- Garratt MP, Leather SR, Wright DJ. 2010b. Tritrophic effects of organic and conventional fertilizers on a cereal aphid parasitoid system. Entomol. Exp. Appl. 134: 211-219. [Google Scholar]
- Gill HK, Garg H, Gillett-Kaufman JL. 2024. Featured creatures: Cabbage aphid. University of Florida, USA. Accessed URL: http://entnemdept.ufl.edu/creatures/veg/aphid/Cabbage_aphid.htm. Accessed on June 5, 2024. [Google Scholar]
- Giordanengo P, Brunissen L, Rusterucci C, et al. 2010. Compatible plant-aphid interactions: how aphids manipulate plant responses. Comp. Rend. Biol. 333: 516-523. [Google Scholar]
- Girousse C, Moulia B, Silk W, Bonnemain JL. 2005. Aphid infestation causes different changes in carbon and nitrogen allocation in alfalfa stems as well as different inhibitions of longitudinal and radial expansion. Plant Physiol. 137: 1474-1484. [Google Scholar]
- Graca-Souza AV, Maya-Monteiro C, Paiva-Silva GO, et al. 2006. Adaptations against heme toxicity in blood-feeding arthropods. Insect Biochem. Mol. Biol. 36:322-335. [Google Scholar]
- Hatzig, S, Breuer F, Nesi N, et al. 2018. Hidden effects of seed quality breeding on germination in oilseed rape (Brassica napus L.). Front. Plant Sci. 9: e419. [Google Scholar]
- Hofmann T, Chubanov V, Chen X, Dietz AS, Gudermann T, Montell C. 2010. Drosophila TRPM channel is essential for the control of extracellular magnesium levels. Plos One. 5: e10519. [Google Scholar]
- Hwang JE, Bruyne MD, Warr CG, Burke R. 2014. Copper overload and deficiency both adversely affect the central nervous system of Drosophila. Metallomics 6: 2223-2229 [Google Scholar]
- Ijaz M, Sher A, Sattar A, et al. 2019. Response of canola (Brassica napus L.) to exogenous application of nitrogen, salicylic acid and gibberellic acid under an arid climate. Soil Environ. 38: 90-96. [Google Scholar]
- Imas P. 2013. Potassium: The quality element in crop production. Switzerland: International Potash Institute. [Google Scholar]
- Iwashina T. 2003. Flavonoid function and activity to plants and other organisms. Biol. Sci. Space. 17: 24-44. [Google Scholar]
- Jacobo-Velázquez DA, Cisneros-Zevallos L. 2020. Bioactive phenolics and polyphenols: Current advances and future trends. Int. J. Mol. Sci. 21: e6142. [Google Scholar]
- Javed MW, Hasan MU, Sagheer M, Sahi ST. 2020. Studies on inducer mediated resistance responses against biological fitness of Brevicoryne brassicae (Homoptera: Aphididae) on Brassica napus. Int. J. Agri. Biol. 25:81–88 [Google Scholar]
- Javed MW, Hasan MU, Sagheer M, Sahi ST, Mankin RW. 2022. Foliar and soil treatments of Brassica napus that elicit antibiosis in Brevicoryne brassicae. Agronomy 12:e882 [Google Scholar]
- Javed MW, Hasan MU, Sagheer M, et al. 2025a. Soil amendment–mediated herbivory resistance, crop improvement, and phytoremediation in canola: Physiological defense mechanism and health risk assessment. Plants 14:e1110. [Google Scholar]
- Javed MW, Hasan MU, Sagheer M, Sahi ST. 2025b. Silicon–mediated nutrient deterrence could reduce aphid (Brevicoryne brassicae L.) abundance and improve crop yield by eliciting phenolic defense in canola (Brassica napus L.). J. Soil Sci. Plant Nutr. 25:5395–5409 [Google Scholar]
- Javed MW, Hasan MU, Sagheer M, Sahi ST. 2025c. Type–dependent biostimulant–mediated induction of nutrients and phenolic compounds improved plant tolerance against canola aphids (Brevicoryne brassicae L.). Plant Physiol. Biochem. 224:e109930 [Google Scholar]
- Javed MW, Hussain D, Zubair M, et al. 2025d. Sulphur–mediated plant resistance elicits phenolic defense in canola to reduce aphid reproduction and improve crop yield in canola. Ach. Phytopathol. Plant Prot. 58:389–412 [Google Scholar]
- Javed MW, Hasan MU, Sagheer M, Sahi ST. 2025e. Soil–mediated nutrient enhancement improves seed yield, nutrient physiology, and pest resistance in Brassica napus against Brevicoryne brassicae. J. Crop Improve. 39:317–334 [Google Scholar]
- Javed MW, Hasan MU, Sagheer M, Sahi ST. 2025f. Induced resistance and nutrient deterrence reduce aphid reproduction and improve crop yield, phenolic defense, and economic benefits of canola (Brassica napus L.) in the field. Arthropod-Plant Interact. 19: 48 [Google Scholar]
- Javed MW, Hasan MU, Sagheer M, Sahi ST. 2025g. Sulphur nutrition improves plant growth performance against a specialist herbivore by eliciting phenolic defense and nutrient induction in rapeseed. J Plant Growth Regul. https://doi.org/10.1007/s00344-025-11884-4 [Google Scholar]
- Karthik R, Deka MK, Kalita ASP, Prakash N. 2024a. Impact of foliar application of silicic acid on aphid population growth, gas exchange parameters and yield of rapeseed. Phytoparasitica. 52: 65. [Google Scholar]
- Karthik R, Deka MK, Kalita ASP, Prakash N. 2024b. Effect of foliar application of silicic acid on biological parameters of Lipaphis erysimi (Kaltenbach) and activity of plant defensive enzymes in rapeseed. Int. J. Trop. Insect Sci. 44: 2685–2694. [Google Scholar]
- Khan IA, Ahmad M, Akbar R, et al. 2015. A study on losses due to Brevicoryne brassicae in different Brassica genotypes under screen houses conditions. J. Entomol. Zool. Stud. 3: 16-19. [Google Scholar]
- Khan M, Ulrichs C, Mewis I. 2011. Effect of water stress and aphid herbivory on flavonoids in broccoli (Brassica oleracea var. italica Plenck). J. Appl. Bot. Food Qual. 84: 178-182. [Google Scholar]
- Khandekar S, Leisner S. 2011. Soluble silicon modulates expression of Arabidopsis thaliana genes involved in copper stress. J. Plant Physiol. 168: 699-705. [Google Scholar]
- Khattab H. 2007. The defense mechanism of cabbage plant against phloem-sucking aphid (Brevicoryne brassicae L.). Aust. J. Bas. Appl. Sci. 1: 56-62. [Google Scholar]
- Khoshfarman-Borji H, Yali MP, Bozorg-Amirkalaee M. 2020. Induction of resistance against Brevicoryne brassicae by Pseudomonas putida and salicylic acid in canola. Bull. Entomol. Res. 110: 597-610. [Google Scholar]
- Killilea DW, Westropp JL, Shiraki R, et al. 2015. Elemental content of calcium oxalate stones from a canine model of urinary stone disease. Plos One 10: e0128374. [Google Scholar]
- Kirkby E. 2012. Introduction, definition and classification of nutrients. In: P. Marschner, ed. Marschner's mineral nutrition of higher plants. 3rd ed. Academic Press. London, UK pp. 3-5. [Google Scholar]
- Kirkland LS, Chirgwin E, Ward SE, Congdon BS, van Rooyen A, Umina PA. 2023. P450-mediated resistance in Myzus persicae (Sulzer) (Hemiptera: Aphididae) reduces the efficacy of neonicotinoid seed treatments in Brassica napus. Pest Manag. Sci. 79:1851-1859. [Google Scholar]
- Kumar S, Singh Y, Singh S, Singh R. 2017. Physical and biochemical aspects of host plant resistance to mustard aphid, Lipaphis erysimi (Kaltenbach) in rapeseed-mustard. Arth. Plant Interact. 11: 551-559. [Google Scholar]
- Lu ZX, Yu XP, Heong KL, Cui HU. 2007. Effect of nitrogen fertilizer on herbivores and its stimulation to major insect pests in rice. Rice Sci. 14: 56-66. [Google Scholar]
- Łukasik I, Goławska S. 2013. Effect of host plant on levels of reactive oxygen species and antioxidants in the cereal aphids Sitobion avenae and Rhopalosiphum padi. Biochem. System Ecol. 51: 232-239. [Google Scholar]
- Łukasik I, Kornacka A, Goławska S, Sytykiewicz H, Sprawka I, Wójcicka A. 2017. Effects of Acyrtosiphon pisum (Harris) infestation on the hydrogen peroxide content and activity of antioxidant enzymes in Fabaceae plants. Allelopathy J. 40: 143-150. [Google Scholar]
- Maazoun, AM, Hamdi SH, Belhadj F, Jemâa JMB, Messaoud C, Marzouki MN. 2019. Phytochemical profile and insecticidal activity of Agave americana leaf extract towards Sitophilus oryzae (L.) (Coleoptera: Curculionidae). Environ. Sci. Poll. Res. 26: 19468-19480. [Google Scholar]
- Mahmood T, Mustafa HSB, Aftab M, Ali Q, Malik A. 2019. Super canola: newly developed high yielding, lodging and drought tolerant double zero cultivar of rapeseed (Brassica napus L.). Gen. Mol. Res. 18: gmr16039951. [Google Scholar]
- Moran NA. 1992. The evolution of aphid life cycles. Ann. Rev. Entomol. 37: 321-348. [Google Scholar]
- Mousavi AS. 2017. Resistance of four canola genotypes against cabbage aphid Brevicoryne brassicae (L.). J. Ir. Plant Prot. Res. 31: 191-198. [Google Scholar]
- Mpumi N, Machunda RS, Mtei KM, Ndakidemi PA. 2020. Selected insect pests of economic importance to Brassica oleracea, their control strategies and the potential threat to environmental pollution in Africa. Sustainability. 12: e3824. [Google Scholar]
- Myers SW, Gratton C. 2006. Influence of potassium fertility on soybean aphid, Aphis glycines Matsumura (Hemiptera: Aphididae), population dynamics at a field and regional scale. Environ. Entomol. 35: 219-227. [Google Scholar]
- Nasab RS, Yali MP, Bozorg-Amirkalaee M. 2019. Effects of humic acid and plant growth-promoting rhizobacteria (PGPR) on induced resistance of canola to Brevicoryne brassicae L. Bull. Entomol. Res. 109: 479-489. [Google Scholar]
- Oberdörster E, Clay MA, Cottam DM, Wilmot FA, McLachlan JA, Milner MJ. 2001. Common phytochemicals are ecdysteroid agonists and antagonists: A possible evolutionary link between vertebrate and invertebrate steroid hormones. J. Ster. Biochem. Mol. Biol. 77: 229-238. [Google Scholar]
- Okumoto S, Pilot G. 2011. Amino acid export in plants: A missing link in nitrogen cycling. Mol. Plant 4: 453-463. [Google Scholar]
- Pajar JA, Otto P, Leonar AL, Döll S, van Dam NM. 2024. Dual nematode infection in Brassica nigra affects shoot metabolome and aphid survival in distinct contrast to single-species infection. J. Exp. Bot. 75: 7317–7336. [Google Scholar]
- Pan J, Farhan M, Liu X, Li H, Yin Z, Zhang S. 2026. Adaptive divergence of three Aphis gossypii haplotypes on leguminous hosts. Pest Manag. Sci. 82:169-182. [Google Scholar]
- Pavlovic J, Samardzic J, Maksimović V, et al. 2013. Silicon alleviates iron deficiency in cucumber by promoting mobilization of iron in the root apoplast. New Phytol. 199: 1096-1107. [Google Scholar]
- Qayyum A, Sarfraz RA, Ashraf A, Adil S. 2016. Phenolic composition and biological (anti diabetic and antioxidant) activities of different solvent extracts of an endemic plant (Heliotropium strigosum). J. Chil. Chem. Soc. 61: 2828-2831. [Google Scholar]
- Razaq M, Mehmood A, Aslam M, Ismail M, Afzal M, Shad SA. 2011. Losses in yield and yield components caused by aphids to late sown Brassica napus L., Brassica juncea L. and Brassica carrinata A. Braun at Multan, Punjab (Pakistan). Pak. J. Bot. 43: 319-324. [Google Scholar]
- Razaq M, Farooq M, Abbass G, et al. 2016. Is reduction in yield potential of some brassicaceous species due to aphid infestation associated with the changes in stomatal factors of photosynthesis? Pak. J. Bot. 48: 1665-1671. [Google Scholar]
- Razmjou J, Jafary M, Borzoui E. 2019. Host plant preference and life table of Brevicoryne brassicae (Hemiptera: Aphididae). J. Crop Prot. 8: 201-214. [Google Scholar]
- Rehman H, Iqbal Q, Farooq M, Wahid A, Afzal I, Basra SM. 2013. Sulphur application improves the growth, seed yield and oil quality of canola. Acta Physiol. Plant. 35: 2999-3006. [Google Scholar]
- Riedell WE, Osborne SL, Jaradat AA. 2007. Crop mineral nutrient and yield responses to aphids or barley yellow dwarf virus in spring wheat and oat. Crop Sci. 47: 1553-1560. [Google Scholar]
- Rowntree JK, McVennon A, Preziosi RF. 2010. Plant genotype mediates the effects of nutrients on aphids. Oecologia. 163: 675-679. [Google Scholar]
- Roy N, Ghosh GK. 2020. Enhancing nutrient availability, yield and quality of safflower (Carthamus tinctorius L.) through zinc and sulphur in Alfisol. J. Pharma. Phytochem. 9: 693-697. [Google Scholar]
- Safar-Noori M, Assaha DVM, Saneoka H. 2018. Effect of salicylic acid and potassium application on yield and grain nutritional quality of wheat under drought stress condition. Cer. Res. Com. 46: 558-568. [Google Scholar]
- Saikia, SL, Rai GK, Salgotra R, Rai S, Singh M, Rai P. 2018. Erucic acid and glucosinolate variability in Brassica juncea L. Int. J. Chem. Stud. 6: 1223-1226. [Google Scholar]
- Samanta A, Das G, Das SK. 2011. Roles of flavonoids in plants. Carbon. 100: 12-35. [Google Scholar]
- Satar S, Kersting U, Ulusoy MR. 2005. Temperature dependent life history traits of Brevicoryne brassicae (L.) (Hom: Aphididae) on white cabbage. Turk. J. Agric. Forest 29: 341-346. [Google Scholar]
- Shah FM, Razaq M, Ali Q, Shad SA, Aslam M, Hardy IC. 2019. Field evaluation of synthetic and neem-derived alternative insecticides in developing action thresholds against cauliflower pests. Sci. Rep. 9: e7684. [Google Scholar]
- Shahrokhi M, Yali MP, Bozorg-Amirkalaee M. 2024. Role of exogenous elicitors in canola plant defense against cabbage aphid by regulating physiological balance and secondary metabolite biosynthesis. J. Agric. Sci. Technol. 26: 165-176 [Google Scholar]
- Shen J, Yuan L, Zhang J, Li H, Bai Z, Chen X, Zhang W, Zhang F. 2011. Phosphorus dynamics: from soil to plant. Plant Physiol. 156: 997-1005. [CrossRef] [Google Scholar]
- Singh V, Sood AK. 2017. Plant nutrition: A tool for the management of hemipteran insect-pests—a review. Agric. Rev. 38: 260-270. [Google Scholar]
- Smith CM, Boyko EV. 2007. The molecular bases of plant resistance and defense responses to aphid feeding: current status. Entomol. Exp. Appl. 122: 1-16. [Google Scholar]
- Soh BSB, Kekeunou S, Nanga SN, Dongmo M, Rachid H. 2018. Effect of temperature on the biological parameters of the cabbage aphid Brevicoryne brassicae. Ecol. Evol. 8: 11819-11832. [Google Scholar]
- Sosa ME, Guerreiro E, Giordano OS, Tonn CE. 2000. Bioactivity of flavonoids against Tenebrio molitor (Coleoptera: Tenebrionidae) larvae. Rev. Soc. Entomol. Argen. 59: 179-184. [Google Scholar]
- Spilioti E, Jaakkola M, Tolonen T, et al. 2014. Phenolic acid composition, antiatherogenic and anticancer potential of honeys derived from various regions in Greece. Plos One. 9: e94860. [Google Scholar]
- Syeed S, Anjum NA, Nazar R, Iqbal N, Masood A, Khan NA. 2011. Salicylic acid-mediated changes in photosynthesis, nutrients content and antioxidant metabolism in two mustard (Brassica juncea L.) cultivars differing in salt tolerance. Acta Physiol. Plant 33: 877-886. [Google Scholar]
- Sytykiewicz H, Chrzanowski G, Czerniewicz P, et al. 2014. Expression profiling of selected glutathione transferase genes in Zea mays (L.) seedlings infested with cereal aphids. Plos One 9: e111863. [Google Scholar]
- Tajik S, Zarinkamar F, Soltani BM, and Nazari M. 2019. Induction of phenolic and flavonoid compounds in leaves of saffron (Crocus sativus L.) by salicylic acid. Sci. Hort. 257: e108751. [Google Scholar]
- Tao L, Hunter MD. 2012. Does anthropogenic nitrogen deposition induce phosphorus limitation in herbivorous insects? Glob. Change Biol. 18: 1843-1853. [Google Scholar]
- Torres-Olivar V, Villegas-Torres OG, Domínguez-Patino ML, et al. 2014. Role of nitrogen and nutrients in crop nutrition. J. Agric. Sci. Technol. B 4: 29-37. [Google Scholar]
- Tosh CR, Powell G, Holmes ND, Hardie J. 2003. Reproductive response of generalist and specialist aphid morphs with the same genotype to plant secondary compounds and amino acids. J. Insect Physiol. 49: 1173-1182. [Google Scholar]
- Trumper S, Simpson SJ. 1993. Regulation of salt intake by nymphs of Locusta migratoria. J. Insect Physiol. 39: 857-864. [Google Scholar]
- Tsao R. 2010. Chemistry and biochemistry of dietary polyphenols. Nutrients 2: 1231-1246. [Google Scholar]
- Tsuge K, Chen R, Yoshioka Y, Motoki S. 2020. Assessment of genetic diversity based on phenotypic traits and microsatellite markers in Brassica napus L. Landrace in the Kanto Region, Japan (Norabona). Hort. J. 89: 12-21. [Google Scholar]
- Uchida R. 2000. Essential nutrients for plant growth: Nutrient functions and deficiency symptoms. In: Silva JA, Uchida R, eds. Plant Nutrient Management in Hawaii's Soils, Approaches for Tropical and Subtropical Agriculture. College of Tropical Agriculture and Human Resources, University of Hawaii at Manoa. pp. 31-55. [Google Scholar]
- Vendramim JD, Guzzo EC. 2012. Plant resistance and insect bioecology and nutrition. In: Panizzi AR, Parra JRP, eds. Insect Bioecology and Nutrition for Integrated Pest Management, 1st ed. CRC Press. Boca Raton pp. 657-686. [Google Scholar]
- Vuolo MM, Lima VS, and Junior MRM. 2019. Phenolic compounds: Structure, classification, and antioxidant power. In: Bioactive compounds. Woodhead Publishing. Duxford, UK pp. 33-50. [Google Scholar]
- Walter AJ, Difonzo CD. 2014. Soil potassium deficiency affects soybean phloem nitrogen and soybean aphid populations. Environ. Entomol. 36: 26-33. [Google Scholar]
- Wang C, Tian B, Yu Z, Ding J. 2020. Effect of different combinations of phosphorus and nitrogen fertilization on arbuscular mycorrhizal fungi and aphids in wheat. Insects 11: e11060365. [Google Scholar]
- Wang Z, Zhao Z, Cheng X, Liu S, Wei Q, Scott IM. 2016. Conifer flavonoid compounds inhibit detoxification enzymes and synergize insecticides. Pest. Biochem. Physiol. 127: 1-7. [Google Scholar]
- Welter KC, Martins CMMR, de Palma ASV, et al. 2016. Canola oil in lactating dairy cow diets reduces milk saturated fatty acids and improves its omega-3 and oleic fatty acid content. Plos One. 11: e151876. [Google Scholar]
- Wessing A, Zierold K. 1992. Metal-salt feeding causes alterations in concretions in Drosophila larvae Malpighian tubules as revealed by X-ray microanalysis. J. Insect Physiol. 389: 623-632. [Google Scholar]
- White PJ, Brown P. 2010. Plant nutrition for sustainable development and global health. Ann. Bot. 105: 1073-1080. [Google Scholar]
- White PJ, George TS, Dupuy LX, et al. 2013. Root traits for infertile soils. Front. Plant Sci. 4: e193. [Google Scholar]
- Will T, Tjallingii WF, Thönnessen A, van Bel AJ. 2007. Molecular sabotage of plant defense by aphid saliva. Proc. Nat. Acad. Sci. U.S.A. 104: 10536-10541. [Google Scholar]
- Williamson G, Clifford MN. 2010. Colonic metabolites of berry polyphenols: The missing link to biological activity? Br. J. Nutr. 104: 48-66. [Google Scholar]
- Wolko J, Dobrzycka A, Bocianowski J, Bartkowiak-Broda I. 2019. Estimation of heterosis for yield-related traits for single cross and three-way cross hybrids of oilseed rape (Brassica napus L.). Euphytica. 215: e156. [Google Scholar]
- Xiao L, Carrillo J, Siemann E, Ding J. 2019. Herbivore-specific induction of indirect and direct defensive responses in leaves and roots. AoB Plants 11: e003.. [Google Scholar]
- Yang L, Han Y, Li P, Wen L, Hou M. 2017. Silicon amendment to rice plants impairs sucking behaviors and population growth in the phloem feeder Nilaparvata lugens (Hemiptera: Delphacidae). Sci. Rep. 7: e1101. [Google Scholar]
- Yousafi Q, Saleem S. 2020. Benefit-cost ratio evaluation for management of Brevicoryne brassicae L. in canola (Brassica napus L.) crop production. Phytoparasitica. 48: 673-683. [Google Scholar]
- Zaheer IE, Ali S, Rizwan M, et al. 2015. Citric acid assisted phytoremediation of copper by Brassica napus L. Ecotoxicol. Environ. Saf. 120: 310-317. [Google Scholar]
- Zhou F, Chen C, Kong L, et al. 2022. Genetic diversity analysis reveals potential of the green peach aphid (Myzus persicae) resistance in Ethiopian mustard. Int. J. Mol. Sci. 23: 13736. [Google Scholar]
Cite this article as: Muhammad Wajid Javed. 2026. Deciphering canola- aphid interaction: A review on the role of hostplant nutrients and phenolic compounds. OCL 33: 13. https://doi.org/10.1051/ocl/2026006
All Tables
Recent studies on nutrients in aphid-canola interaction along with their major findings.
Categories, sub-types, specific compounds, and the significance of major classes of phenolic compounds in canola.
All Figures
 |
Fig 1 Canola (Brassica napus L.) plant 75 days after sowing (photo by Muhammad Wajid Javed). |
| In the text | |
 |
Fig. 2 (a) Sexual mode of reproduction, (b) asexual mode of reproduction, and (c) general biology of aphids in canola. |
| In the text | |
 |
Fig 3 Infested and non-infested parts of canola (Brassica napus). (a) Aphid-infested inflorescence, (b) aphid-infested pods, (c) a non-infested flower on the left and infested flowers on right side, and (d) aphid-infested pods on the left and a non-infested pod on the right side. Photo courtesy by Muhammad Wajid Javed. |
| In the text | |
 |
Fig 4 Role of primary and secondary macronutrients along with micronutrients for aphids. |
| In the text | |
 |
Fig 5 Nutrient flux-mediated growth stages of canola where aphids can cause significant damage. (a) Flowering, (b) blooming, and (c) pod formation. |
| In the text | |
 |
Fig 6 Categorical role of macronutrients (primary and secondary) and micronutrients for canola. |
| In the text | |
 |
Fig 7 Biosynthesis of hydroxybenzoic and hydroxycinnamic acid from amino acids tyrosine and phenylalanine, using tyrosine ammonia lyase (TAL), L-phenylalanine ammonia-lyase (PAL), and cinnamic acid 4-hydroxylase (CAH) enzymes. NH3 and CH3 show deamination and methylation, respectively. Figure courtesy by Muhammad Wajid Javed. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.




